Search results
Search for "C–H bond activation" in Full Text gives 53 result(s) in Beilstein Journal of Organic Chemistry.
Mono or double Pd-catalyzed C–H bond functionalization for the annulative π-extension of 1,8-dibromonaphthalene: a one pot access to fluoranthene derivatives
- Nahed Ketata,
- Linhao Liu,
- Ridha Ben Salem and
- Henri Doucet
Beilstein J. Org. Chem. 2024, 20, 427–435, doi:10.3762/bjoc.20.37
- palladium-catalyzed direct intermolecular arylation, followed by a direct intramolecular arylation step. As the C–H bond activation of several benzene derivatives remains very challenging, the preparation of fluoranthenes from 1,8-dibromonaphthalene via Suzuki coupling followed by intramolecular C–H
- reported [39][40]. The synthesis of fluoranthenes from 1,8-dibromonaphthalene via a double C–H bond activation of the arene used as coupling partner remains limited to specific arenes featuring an activated C–H bond. Consequently, we also investigated the access to fluoranthenes from 1,8-dibromonaphthalene
- , carbonyl or nitrile. Using the appropriate arenes, this synthetic route enables the desired functional groups to be introduced at positions 7 to 10 of fluoranthenes. The second method involves a Suzuki coupling followed by an intramolecular C–H bond activation step, and provides access to specific

Graphical Abstract

Figure 1: Structure of fluoranthene.

Scheme 1: Pd-catalyzed access to fluoranthenes.

Scheme 2: Scope of the Pd-catalyzed direct arylation reaction of arenes with 1,8-dibromonaphthalene.

Scheme 3: Scope of the Pd-catalyzed direct arylation reaction of 2,5-substituted heteroarenes with 1,8-dibrom...

Scheme 4: Scope of the Pd-catalyzed Suzuki reaction followed by direct arylation of arylboronic acids with 1,...

Scheme 5: Attempted reaction of 1-naphthylboronic acid with 1,2-dihalobenzenes.

Scheme 6: Pd-catalyzed Heck reaction followed by direct arylation of 1,1-diphenylethylene with 1,2-dihalobenz...
Lewis acid-promoted direct synthesis of isoxazole derivatives
- Dengxu Qiu,
- Chenhui Jiang,
- Pan Gao and
- Yu Yuan
Beilstein J. Org. Chem. 2023, 19, 1562–1567, doi:10.3762/bjoc.19.113
- the Lewis acid to realize the sp3 C–H-bond activation of nitrogen heterocycles to synthesize isoxazole derivatives. Results and Discussion At the outset of this study, we chose the reaction of 2-methylquinoline (2a) with phenylacetylene (1a) in the presence of AlCl3 (3 equiv) and sodium nitrite (10
- oxide E [23], which can be converted to the desired isoxazole with 1a through a 1,3-dipolar cycloaddition. Conclusion In conclusion, we have developed an efficient and concise synthesis of isoxazole nitrogen heterocycles by direct C–H-bond activation of methyl heteroaromatics. The method avoids using

Graphical Abstract

Figure 1: Natural products and drug molecules containing isoxazole moieties.

Scheme 1: Traditional methods for the synthesis of isoxazoles and the current approach.

Scheme 2: Reaction scope of alkynes. Conditions: 1 (0.1 mmol, 1 equiv), 2a (0.2 mmol, 2 equiv), AlCl3 (0.3 mm...

Figure 2: Crystal structure of 3i.

Scheme 3: Reaction substrate scope of quinolines. Conditions: 1a (0.1 mmol, 1 equiv), 2 (0.2 mmol, 2 equiv), ...

Scheme 4: Gram scale reaction.

Scheme 5: Control experiments and possible reaction mechanism.
Non-noble metal-catalyzed cross-dehydrogenation coupling (CDC) involving ether α-C(sp3)–H to construct C–C bonds
- Hui Yu and
- Feng Xu
Beilstein J. Org. Chem. 2023, 19, 1259–1288, doi:10.3762/bjoc.19.94
- overcome the shortcomings of the above coupling reactions, organic chemists have envisaged the construction of C–C bonds directly through C–H bond activation [5]. Fortunately, scientists have used various transition metals as catalysts to realize the activation of various types of C–H bonds, and have

Graphical Abstract

Scheme 1: Research progress of coupling reactions and active compounds containing α-C(sp3)-functionalized eth...

Scheme 2: Transition-metal-catalyzed CDC pathways.

Scheme 3: CDC of active methylene compounds in the α-C(sp3) position of ethers.

Scheme 4: InCl3/Cu(OTf)2/NHPI co-catalyzed CDC reaction.

Scheme 5: CDC of cyclic benzyl ethers with aldehydes.

Scheme 6: Cu-catalyzed CDC of (a) unactivated C(sp3)–H ethers with simple ketones and (b) double C(sp3)−H fun...

Scheme 7: Cu-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 8: Cu-catalyzed synthesis of chiral 2-substituted tetrahydropyrans.

Scheme 9: CDC of thiazole with cyclic ethers.

Scheme 10: Cu(I)-catalyzed oxidative alkenylation of simple ethers.

Scheme 11: Cross-dehydrogenation coupling of isochroman C(sp3)–H bonds with anisole C(sp2)–H bonds.

Scheme 12: Pd(OAc)2/Cu(OTf)2-catalyzed arylation of α-C(sp3)–H bonds of ethers.

Scheme 13: Cu-catalyzed C(sp3)–H/C(sp2)–H activation strategies to construct C(sp3)–C(sp2) bonds.

Scheme 14: Cu(I)-catalyzed C(sp2)–H alkylation.

Scheme 15: Cu-catalyzed C(sp3)–H/C(sp)–H activation to construct C(sp3)–C(sp) bonds (H2BIP: 2,6-bis(benzimidaz...

Scheme 16: Fe-catalyzed CDC reaction pathways.

Scheme 17: Fe2(CO)9-catalyzed functionalization of C–H bonds.

Scheme 18: Ligand-promoted Fe-catalyzed CDC reaction of N-methylaniline with ethers.

Scheme 19: Fe-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 20: Fe-catalyzed hydroalkylation of α,β-unsaturated ketones with ethers.

Scheme 21: Solvent-free Fe(NO3)3-catalyzed CDC of C(sp3)–H/C(sp2)–H bonds.

Scheme 22: Alkylation of disulfide compounds to afford tetrasubstituted alkenes.

Scheme 23: Fe-catalyzed formation of 1,1-bis-indolylmethane derivatives.

Scheme 24: Alkylation of coumarins and flavonoids.

Scheme 25: Direct CDC α-arylation of azoles with ethers.

Scheme 26: CDC of terminal alkynes with C(sp3)–H bonds adjacent to oxygen, sulfur or nitrogen atoms.

Scheme 27: Alkylation of terminal alkynes.

Scheme 28: Co-catalyzed functionalization of glycine esters.

Scheme 29: Co-catalyzed construction of C(sp2)–C(sp3) bonds.

Scheme 30: Co-catalyzed CDC of imidazo[1,2-a]pyridines with isochroman.

Scheme 31: Co-catalyzed C–H alkylation of (benz)oxazoles with ethers.

Scheme 32: Cobalt-catalyzed CDC between unactivated C(sp2)–H and C(sp3)–H bonds.

Scheme 33: MnO2-catalyzed CDC of the inactive C(sp3)-H.

Scheme 34: Oxidative cross-coupling of ethers with enamides.

Scheme 35: Ni(II)-catalyzed CDC of indoles with 1,4-dioxane.

Scheme 36: Chemo- and regioselective ortho- or para-alkylation of pyridines.

Scheme 37: Asymmetric CDC of 3,6-dihydro-2H-pyrans with aldehydes.

Scheme 38: CDC of heterocyclic aromatics with ethers.

Scheme 39: Indium-catalyzed alkylation of DHPs with 1,3-dicarbonyl compounds.

Scheme 40: Rare earth-metal-catalyzed CDC reaction.

Scheme 41: Visible-light-driven CDC of cycloalkanes with benzazoles.

Scheme 42: Photoinduced alkylation of quinoline with cyclic ethers.

Scheme 43: Photocatalyzed CDC reactions between α-C(sp3)–H bonds of ethers and C(sp2)–H bonds of aromatics.
Pyridine C(sp2)–H bond functionalization under transition-metal and rare earth metal catalysis
- Haritha Sindhe,
- Malladi Mounika Reddy,
- Karthikeyan Rajkumar,
- Akshay Kamble,
- Amardeep Singh,
- Anand Kumar and
- Satyasheel Sharma
Beilstein J. Org. Chem. 2023, 19, 820–863, doi:10.3762/bjoc.19.62
- also found to have a higher catalytic activity for the ortho-alkylation of pyridines with styrenes to give the linear alkylated products (5b,c, Scheme 2). Further, the authors proposed that the C–H bond activation could be the rate limiting step based on kinetic isotope experiments (KIE). The proposed

Graphical Abstract

Figure 1: Representative examples of bioactive natural products and FDA-approved drugs containing a pyridine ...

Scheme 1: Classical and traditional methods for the synthesis of functionalized pyridines.

Scheme 2: Rare earth metal (Ln)-catalyzed pyridine C–H alkylation.

Scheme 3: Pd-catalyzed C–H alkylation of pyridine N-oxide.

Scheme 4: CuI-catalyzed C–H alkylation of N-iminopyridinium ylides with tosylhydrazones (A) and a plausible r...

Scheme 5: Zirconium complex-catalyzed pyridine C–H alkylation.

Scheme 6: Rare earth metal-catalyzed pyridine C–H alkylation with nonpolar unsaturated substrates.

Scheme 7: Heterobimetallic Rh–Al complex-catalyzed ortho-C–H monoalkylation of pyridines.

Scheme 8: Mono(phosphinoamido)-rare earth complex-catalyzed pyridine C–H alkylation.

Scheme 9: Rhodium-catalyzed pyridine C–H alkylation with acrylates and acrylamides.

Scheme 10: Ni–Al bimetallic system-catalyzed pyridine C–H alkylation.

Scheme 11: Iridium-catalyzed pyridine C–H alkylation.

Scheme 12: para-C(sp2)–H Alkylation of pyridines with alkenes.

Scheme 13: Enantioselective pyridine C–H alkylation.

Scheme 14: Pd-catalyzed C2-olefination of pyridines.

Scheme 15: Ru-catalyzed C-6 (C-2)-propenylation of 2-arylated pyridines.

Scheme 16: C–H addition of allenes to pyridines catalyzed by half-sandwich Sc metal complex.

Scheme 17: Pd-catalyzed stereodivergent synthesis of alkenylated pyridines.

Scheme 18: Pd-catalyzed ligand-promoted selective C3-olefination of pyridines.

Scheme 19: Mono-N-protected amino acids in Pd-catalyzed C3-alkenylation of pyridines.

Scheme 20: Amide-directed and rhodium-catalyzed C3-alkenylation of pyridines.

Scheme 21: Bimetallic Ni–Al-catalyzed para-selective alkenylation of pyridine.

Scheme 22: Arylboronic ester-assisted pyridine direct C–H arylation.

Scheme 23: Pd-catalyzed C–H arylation/benzylation with toluene.

Scheme 24: Pd-catalyzed pyridine C–H arylation with potassium aryl- and heteroaryltrifluoroborates.

Scheme 25: Transient activator strategy in pyridine C–H biarylation.

Scheme 26: Ligand-promoted C3-arylation of pyridine.

Scheme 27: Pd-catalyzed arylation of nicotinic and isonicotinic acids.

Scheme 28: Iron-catalyzed and imine-directed C–H arylation of pyridines.

Scheme 29: Pd–(bipy-6-OH) cooperative system-mediated direct pyridine C3-arylation.

Scheme 30: Pd-catalyzed pyridine N-oxide C–H arylation with heteroarylcarboxylic acids.

Scheme 31: Pd-catalyzed C–H cross-coupling of pyridine N-oxides with five-membered heterocycles.

Scheme 32: Cu-catalyzed dehydrative biaryl coupling of azine(pyridine) N-oxides and oxazoles.

Scheme 33: Rh(III)-catalyzed cross dehydrogenative C3-heteroarylation of pyridines.

Scheme 34: Pd-catalyzed C3-selective arylation of pyridines.

Scheme 35: Rhodium-catalyzed oxidative C–H annulation of pyridines to quinolines.

Scheme 36: Rhodium-catalyzed and NHC-directed C–H annulation of pyridine.

Scheme 37: Ni/NHC-catalyzed regio- and enantioselective C–H cyclization of pyridines.

Scheme 38: Rare earth metal-catalyzed intramolecular C–H cyclization of pyridine to azaindolines.

Scheme 39: Rh-catalyzed alkenylation of bipyridine with terminal silylacetylenes.

Scheme 40: Rollover cyclometallation in Rh-catalyzed pyridine C–H functionalization.

Scheme 41: Rollover pathway in Rh-catalyzed C–H functionalization of N,N,N-tridentate chelating compounds.

Scheme 42: Pd-catalyzed rollover pathway in bipyridine-6-carboxamides C–H arylation.

Scheme 43: Rh-catalyzed C3-acylmethylation of bipyridine-6-carboxamides with sulfoxonium ylides.

Scheme 44: Rh-catalyzed C–H functionalization of bipyridines with alkynes.

Scheme 45: Rh-catalyzed C–H acylmethylation and annulation of bipyridine with sulfoxonium ylides.

Scheme 46: Iridium-catalyzed C4-borylation of pyridines.

Scheme 47: C3-Borylation of pyridines.

Scheme 48: Pd-catalyzed regioselective synthesis of silylated dihydropyridines.
Enolates ambushed – asymmetric tandem conjugate addition and subsequent enolate trapping with conventional and less traditional electrophiles
- Péter Kisszékelyi and
- Radovan Šebesta
Beilstein J. Org. Chem. 2023, 19, 593–634, doi:10.3762/bjoc.19.44

Graphical Abstract

Scheme 1: General scheme depicting tandem reactions based on an asymmetric conjugate addition followed by an ...

Scheme 2: Cu-catalyzed tandem conjugate addition of R2Zn/aldol reaction with chiral acetals.

Scheme 3: Cu-catalyzed asymmetric desymmetrization of cyclopentene-1,3-diones using a tandem conjugate additi...

Scheme 4: Stereocontrolled assembly of dialkylzincs, cyclic enones, and sulfinylimines utilizing a Cu-catalyz...

Scheme 5: Cu-catalyzed tandem conjugate addition/Mannich reaction (A). Access to chiral isoindolinones and tr...

Scheme 6: Cu-catalyzed tandem conjugate addition/nitro-Mannich reaction (A) with syn–anti or syn–syn selectiv...

Figure 1: Various chiral ligands utilized for the tandem conjugate addition/Michael reaction sequences.

Scheme 7: Cu-catalyzed tandem conjugate addition/Michael reaction: side-product formation with chalcone (A) a...

Scheme 8: Zn enolate trapping using allyl iodides (A), Stork–Jung vinylsilane reagents (B), and allyl bromide...

Scheme 9: Cu-catalyzed tandem conjugate addition/acylation through Li R2Zn enolate (A). A four-component coup...

Scheme 10: Selected examples for the Cu-catalyzed tandem conjugate addition/trifluoromethylthiolation sequence....

Scheme 11: Zn enolates trapped by vinyloxiranes: synthesis of allylic alcohols.

Scheme 12: Stereoselective cyclopropanation of Mg enolates formed by ACA of Grignard reagents to chlorocrotona...

Scheme 13: Domino aldol reactions of Mg enolates formed from coumarin and chromone.

Scheme 14: Oxidative coupling of ACA-produced Mg enolates.

Scheme 15: Tandem ACA of Grignard reagents to enones and Mannich reaction.

Scheme 16: Diastereodivergent Mannich reaction of Mg enolates with differently N-protected imines.

Scheme 17: Tandem Grignard–ACA–Mannich using Taddol-based phosphine-phosphite ligands.

Scheme 18: Tandem reaction of Mg enolates with aminomethylating reagents.

Scheme 19: Tandem reaction composed of Grignard ACA to alkynyl enones.

Scheme 20: Rh/Cu-catalyzed tandem reaction of diazo enoates leading to cyclobutanes.

Scheme 21: Tandem Grignard-ACA of cyclopentenones and alkylation of enolates.

Scheme 22: Tandem ACA of Grignard reagents followed by enolate trapping reaction with onium compounds.

Scheme 23: Mg enolates generated from unsaturated lactones in reaction with activated alkenes.

Scheme 24: Lewis acid mediated ACA to amides and SN2 cyclization of a Br-appended enolate.

Scheme 25: Trapping reactions of aza-enolates with Michael acceptors.

Scheme 26: Si enolates generated by TMSOTf-mediated ACA of Grignard reagents and enolate trapping reaction wit...

Scheme 27: Trapping reactions of enolates generated from alkenyl heterocycles (A) and carboxylic acids (B) wit...

Scheme 28: Reactions of heterocyclic Mg enolates with onium compounds.

Scheme 29: Synthetic transformations of cycloheptatrienyl and benzodithiolyl substituents.

Scheme 30: Aminomethylation of Al enolates generated by ACA of trialkylaluminum reagents.

Scheme 31: Trapping reactions of enolates with activated alkenes.

Scheme 32: Alkynylation of racemic aluminum or magnesium enolates.

Scheme 33: Trapping reactions of Zr enolates generated by Cu-ACA of organozirconium reagents.

Scheme 34: Chloromethylation of Zr enolates using the Vilsmeier–Haack reagent.

Scheme 35: Tandem conjugate borylation with subsequent protonation or enolate trapping by an electrophile.

Scheme 36: Tandem conjugate borylation/aldol reaction of cyclohexenones.

Scheme 37: Selected examples for the tandem asymmetric borylation/intramolecular aldol reaction; synthesis of ...

Scheme 38: Cu-catalyzed tandem methylborylation of α,β-unsaturated phosphine oxide in the presence of (R,Sp)-J...

Scheme 39: Cu-catalyzed tandem transannular conjugated borylation/aldol cyclization of macrocycles containing ...

Scheme 40: Stereoselective tandem conjugate borylation/Mannich cyclization: selected examples (A) and a multi-...

Scheme 41: Some examples of Cu-catalyzed asymmetric tandem borylation/aldol cyclization (A). Application to di...

Scheme 42: Atropisomeric P,N-ligands used in tandem conjugate borylation/aldol cyclization sequence.

Scheme 43: Selected examples for the enantioselective Cu-catalyzed borylation/intramolecular Michael addition ...

Scheme 44: Selected examples for the preparation of enantioenriched spiroindanes using a Cu-catalyzed tandem c...

Scheme 45: Enantioselective conjugate borylation of cyclobutene-1-carboxylic acid diphenylmethyl ester 175 wit...

Scheme 46: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 47: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 48: Cu-catalyzed tandem conjugate silylation/aldol condensation. The diastereoselectivity is controlled...

Scheme 49: Chiral Ru-catalyzed three-component coupling reaction.

Scheme 50: Rh-Phebox complex-catalyzed reductive cyclization and subsequent reaction with Michael acceptors th...

Scheme 51: Rh-catalyzed tandem asymmetric conjugate alkynylation/aldol reaction (A) and subsequent spiro-cycli...

Scheme 52: Rh-bod complex-catalyzed tandem asymmetric conjugate arylation/intramolecular aldol addition (A). S...

Scheme 53: Co-catalyzed C–H-bond activation/asymmetric conjugate addition/aldol reaction.

Scheme 54: (Diisopinocampheyl)borane-promoted 1,4-hydroboration of α,β-unsaturated morpholine carboxamides and...

Figure 2: Some examples of total syntheses that have been recently reviewed.

Scheme 55: Stereoselective synthesis of antimalarial prodrug (+)-artemisinin utilizing a tandem conjugate addi...

Scheme 56: Amphilectane and serrulatane diterpenoids: preparation of chiral starting material via asymmetric t...

Scheme 57: Various asymmetric syntheses of pleuromutilin and related compounds based on a tandem conjugate add...

Scheme 58: Total synthesis of glaucocalyxin A utilizing a tandem conjugate addition/acylation reaction sequenc...

Scheme 59: Installation of the exocyclic double bond using a tandem conjugate addition/aminomethylation sequen...

Scheme 60: Synthesis of the taxol core using a tandem conjugate addition/enolate trapping sequence with Vilsme...

Scheme 61: Synthesis of the tricyclic core of 12-epi-JBIR-23/24 utilizing a Rh-catalyzed asymmetric conjugate ...

Scheme 62: Total synthesis of (−)-peyssonoside A utilizing a Cu-catalyzed enantioselective tandem conjugate ad...
Transition-metal-catalyzed C–H bond activation as a sustainable strategy for the synthesis of fluorinated molecules: an overview
- Louis Monsigny,
- Floriane Doche and
- Tatiana Besset
Beilstein J. Org. Chem. 2023, 19, 448–473, doi:10.3762/bjoc.19.35
- functionalization of C(sp2) and C(sp3) centers with SCF3, SeCF3, or OCH2CF3 groups among others, by C–H bond activation. The scope and limitations of these transformations are discussed in this review. Keywords: C–H bond activation; emergent fluorinated groups; homogeneous catalysis; organofluorine chemistry
- retrosynthetic disconnections. In this context, a strong interest from the scientific community was shown towards the challenging synthesis of fluorinated molecules by transition-metal-catalyzed C–H bond activation [44][45][46][47][48][49][50], allowing the functionalization of complex molecules and even for
- (e.g., SCF3, SeCF3, SCF2CO2Et, OCH2CF3) by transition-metal-catalyzed C–H bond activation (Scheme 1). The review will be organized in two main parts, dedicated to the construction of a C–SCF2R/SeCF3 and C–OCH2CF3 bond. This review does not aim to be exhaustive and key examples were carefully chosen to

Graphical Abstract

Scheme 1: Transition-metal-catalyzed C–XRF bond formation by C–H bond activation: an overview.

Scheme 2: Cu(OAc)2-promoted mono- and ditrifluoromethylthiolation of benzamide derivatives derived from 8-ami...

Scheme 3: Trifluoromethylthiolation of azacalix[1]arene[3]pyridines using copper salts and a nucleophilic SCF3...

Scheme 4: Working hypothesis for the palladium-catalyzed C–H trifluoromethylthiolation reaction.

Scheme 5: Trifluoromethylthiolation of 2-arylpyridine derivatives and analogs by means of palladium-catalyzed...

Scheme 6: C(sp2)–SCF3 bond formation by Pd-catalyzed C–H bond activation using AgSCF3 and Selectfluor® as rep...

Scheme 7: Palladium-catalyzed ortho-trifluoromethylthiolation of 2-arylpyridine derivatives reported by the g...

Scheme 8: Palladium-catalyzed ortho-trifluoromethylthiolation of 2-arylpyridine and analogs reported by Anbar...

Scheme 9: Mono- and ditrifluoromethylthiolation of benzamide derivatives derived from 8-aminoquinoline using ...

Scheme 10: Regioselective Cp*Rh(III)-catalyzed directed trifluoromethylthiolation reported by the group of Li [123]...

Scheme 11: Cp*Co(III)-catalyzed ortho-trifluoromethylthiolation of 2-phenylpyridine and 2-phenylpyrimidine der...

Scheme 12: Cp*Co(III)-catalyzed ortho-trifluoromethylthiolation of 2-phenylpyridine and 6-phenylpurine derivat...

Scheme 13: Diastereoselective trifluoromethylthiolation of acrylamide derivatives derived from 8-aminoquinolin...

Scheme 14: C(sp3)–SCF3 bond formation on aliphatic amide derivatives derived from 8-aminoquinoline by palladiu...

Scheme 15: Regio- and diastereoselective difluoromethylthiolation of acrylamides under palladium catalysis rep...

Scheme 16: Palladium-catalyzed (ethoxycarbonyl)difluoromethylthiolation reaction of 2-(hetero)aryl and 2-(α-ar...

Scheme 17: Pd(II)-catalyzed trifluoromethylselenolation of benzamides derived from 5-methoxy-8-aminoquinoline ...

Scheme 18: Pd(II)-catalyzed trifluoromethylselenolation of acrylamide derivatives derived from 5-methoxy-8-ami...

Scheme 19: Transition-metal-catalyzed dehydrogenative 2,2,2-trifluoroethoxylation of (hetero)aromatic derivati...

Scheme 20: Pd(II)-catalyzed ortho-2,2,2-trifluoroethoxylation of N-sulfonylbenzamides reported by the group of...

Scheme 21: Pd(II)-catalyzed selective 2,2,2-trifluoroethoxylation and other fluoroalkoxylations of naphthalene...

Scheme 22: Pd(II)-catalyzed selective ortho-2,2,2-trifluoroethoxylation of benzaldehyde derivatives by means o...

Scheme 23: Pd(II)-catalyzed selective ortho-2,2,2-trifluoroethoxylation (and other fluoroalkoxylations) of ben...

Scheme 24: Pd(II)-catalyzed selective 2,2,2-trifluoroethoxylation of aliphatic amides using a bidentate direct...
Functionalization of imidazole N-oxide: a recent discovery in organic transformations
- Koustav Singha,
- Imran Habib and
- Mossaraf Hossain
Beilstein J. Org. Chem. 2022, 18, 1575–1588, doi:10.3762/bjoc.18.168
- review paper exploring several procedures of C–H bond activation for the functionalization of N-oxides [12] and in 2019, Dongli Li and co-workers analyzed heterocyclic N-oxides with regard to their usefulness in synthesis of organic drug molecules and catalysis [13]. Many review papers have been

Graphical Abstract

Figure 1: Selected imidazole-based bioactive molecules.

Scheme 1: Formation of ethyl 2-cyano-2-(1,3-dihydro-2H-imidazole-2-ylidene)acetate derivatives via [3 + 2] cy...

Scheme 2: C–H/C–Li coupling reaction of 2H-imidazole 1-oxides with pentafluorophenyllithium.

Scheme 3: Transition-metal-free coupling reaction of 2H-imidazole 1-oxides with polyphenols. Reaction conditi...

Scheme 4: Halogenation reaction of 2-unsubstituted imidazole N-oxides using tosyl halogenides.

Scheme 5: Solvent-free chlorination reaction of imidazole N-oxides.

Scheme 6: Multicomponent reaction of imidazole N-oxides 28 with Meldrum’s acid (26) and aldehydes.

Scheme 7: Multicomponent reaction of imidazole N-oxides with CH-acids and aldehydes. Reaction conditions: aTh...

Scheme 8: Three-component condensation reaction of imidazole N-oxides, arylglyoxals, and CH-acids 38 (dimedon...

Scheme 9: Synthesis of imidazole-2-thiones containing cyclohexyl-substituents at 3-position.

Scheme 10: Preparation of optically active derivatives of 3-butoxyimidazole-2-thione.
Mechanochemical halogenation of unsymmetrically substituted azobenzenes
- Dajana Barišić,
- Mario Pajić,
- Ivan Halasz,
- Darko Babić and
- Manda Ćurić
Beilstein J. Org. Chem. 2022, 18, 680–687, doi:10.3762/bjoc.18.69
- dynamics of the formation of cyclopalladated intermediates In-I, LAG reactions of Pd(OAc)2 with L6–8 and TsOH were performed using 25 µL of MeCN as a liquid additive in a molar ratio of 1:1:1 (Ln/Pd(OAc)2/TsOH). In situ Raman monitoring of C–H bond activation was possible for L6 and L7, while in the case

Graphical Abstract

Figure 1: Molecular structures of the monomeric cyclopalladated intermediate and brominated product observed ...

Scheme 1: Halogenation of azobenzenes with strong electron-donating substituents.

Figure 2: a) Two-dimensional (2D) plot of the time-resolved Raman monitoring of NG of L2 (0.50 mmol) with NBS...

Figure 3: Experimental X-ray molecular structure of succinimide product L4-III.

Scheme 2: PdII-catalyzed halogenation of azobenzene and its para-halogenated derivatives.

Figure 4: Experimental X-ray molecular structure of the intermediate I6-I.

Figure 5: a) In situ observation of I6-I during the time-resolved Raman monitoring of LAG of L6 (0.50 mmol) w...
Site-selective reactions mediated by molecular containers
- Rui Wang and
- Yang Yu
Beilstein J. Org. Chem. 2022, 18, 309–324, doi:10.3762/bjoc.18.35
- , directing groups are introduced to the substrates covalently to achieve site-selective C–H bond activation, which prospered greatly in the past decades [7][8][9]. Template regulation is also introduced to locate reactive centers in a noncovalent way through hydrogen bonding [10][11][12]. Even though

Graphical Abstract

Figure 1: Site-selective Diels–Alder reaction of anthracene and phthalimide mediated by aqueous organopalladi...

Figure 2: Site-selective Diels–Alder and [2 + 2]-photoaddition reactions between naphthalene and phthalimide ...

Figure 3: Cage host A-mediated selective 1,4-radical addition of o-quinone 10.

Figure 4: Cyclodextrin-mediated site-selective reductions.

Figure 5: Selective reduction of an α,ω-diazide compound mediated by water-soluble cavitand D.

Figure 6: Selective radical reduction of α,ω-dihalides mediated by water-soluble cavitands E and F.

Figure 7: Site-selective hydrogenation of polyenols mediated by supramolecular encapsulated rhodium catalyst.

Figure 8: Site-selective oxidation of steroids using cyclodextrin as the anchoring template.

Figure 9: Site-selective oxidations of linear diterpenoids with the help of cage host A.

Figure 10: Site-selective monoepoxidation of α,ω-dienes mediated by the water-soluble cavitand host E.

Figure 11: Site-selective ring-opening reaction of epoxides mediated by cavitand I with an inwardly directed c...

Figure 12: Site-selective nucleophilic substitution reaction of allylic chlorides mediated by cage host J.

Figure 13: Site-selective monohydrolysis of α,ω-difunctional compounds using deep water-soluble cavitands.
Recent advances in the syntheses of anthracene derivatives
- Giovanni S. Baviera and
- Paulo M. Donate
Beilstein J. Org. Chem. 2021, 17, 2028–2050, doi:10.3762/bjoc.17.131
- by treatment with PCC/Celite in dichloromethane (DCM). Finally, treatment with a catalytic amount of CF3SO3H provided the corresponding anthracenes 30a–c in good yields (57–75%) [40]. Metal-catalyzed C–H bond activation In 2016, Hong’s group developed a synthetic strategy to generate substituted
- converted to the substituted anthracenes 109 and 110 by varying the acidic workup procedures. In addition, they prepared 9-chloro-10-phenylanthracene (112) in good yield (87%) through diol 111 [59]. Synthesis of substituted benzo[a]anthracene and dibenzoanthracene derivatives Metal-catalyzed C–H bond
- activation In 2009, Liang et al. reported an efficient and highly regioselective route to construct substituted tetracyclic benz[a]anthracene derivatives 115 (Scheme 26) [60]. For this purpose, the authors developed an efficient palladium-catalyzed tandem C–H activation/bis-cyclization reaction of

Graphical Abstract

Figure 1: Examples of anthracene derivatives and their applications.

Scheme 1: Rhodium-catalyzed oxidative coupling reactions of arylboronic acids with internal alkynes.

Scheme 2: Rhodium-catalyzed oxidative benzannulation reactions of 1-adamantoyl-1-naphthylamines with internal...

Scheme 3: Gold/bismuth-catalyzed cyclization of o-alkynyldiarylmethanes.

Scheme 4: [2 + 2 + 2] Cyclotrimerization reactions with alkynes/nitriles in the presence of nickel and cobalt...

Scheme 5: Cobalt-catalyzed [2 + 2 + 2] cyclotrimerization reactions with bis(trimethylsilyl)acetylene (23).

Scheme 6: [2 + 2 + 2] Alkyne-cyclotrimerization reactions catalyzed by a CoCl2·6H2O/Zn reagent.

Scheme 7: Pd(II)-catalyzed sp3 C–H alkenylation of diphenyl carboxylic acids with acrylates.

Scheme 8: Pd(II)-catalyzed sp3 C–H arylation with o-tolualdehydes and aryl iodides.

Scheme 9: Alkylation of arenes with aromatic aldehydes in the presence of acetyl bromide and ZnBr2/SiO2.

Scheme 10: BF3·H2O-catalyzed hydroxyalkylation of arenes with aromatic dialdehyde 44.

Scheme 11: Bi(OTf)3-promoted Friedel–Crafts alkylation of triarylmethanes and aromatic acylals and of arenes a...

Scheme 12: Reduction of anthraquinones by using Zn/pyridine or Zn/NaOH reductive methods.

Scheme 13: Two-step route to novel substituted Indenoanthracenes.

Scheme 14: Synthesis of 1,8-diarylanthracenes through Suzuki–Miyaura coupling reaction in the presence of Pd-P...

Scheme 15: Synthesis of five new substituted anthracenes by using LAH as reducing agent.

Scheme 16: One-pot procedure to synthesize substituted 9,10-dicyanoanthracenes.

Scheme 17: Reduction of bromoanthraquinones with NaBH4 in alkaline medium.

Scheme 18: In(III)-catalyzed reductive-dehydration intramolecular cycloaromatization of 2-benzylic aromatic al...

Scheme 19: Acid-catalyzed cyclization of new O-protected ortho-acetal diarylmethanols.

Scheme 20: Lewis acid-mediated regioselective cyclization of asymmetric diarylmethine dipivalates and diarylme...

Scheme 21: BF3·OEt2/CF3SO3H-mediated cyclodehydration reactions of 2-(arylmethyl)benzaldehydes and 2-(arylmeth...

Scheme 22: Synthesis of 2,3,6,7-anthracenetetracarbonitrile (90) by double Wittig reaction followed by deprote...

Scheme 23: Homo-elongation protocol for the synthesis of substituted acene diesters/dinitriles.

Scheme 24: Synthesis of two new parental BN anthracenes via borylative cyclization.

Scheme 25: Synthesis of substituted anthracenes from a bifunctional organomagnesium alkoxide.

Scheme 26: Palladium-catalyzed tandem C–H activation/bis-cyclization of propargylic carbonates.

Scheme 27: Ruthenium-catalyzed C–H arylation of acetophenone derivatives with arenediboronates.

Scheme 28: Pd-catalyzed intramolecular cyclization of (Z,Z)-p-styrylstilbene derivatives.

Scheme 29: AuCl-catalyzed double cyclization of diiodoethynylterphenyl compounds.

Scheme 30: Iodonium-induced electrophilic cyclization of terphenyl derivatives.

Scheme 31: Oxidative photocyclization of 1,3-distyrylbenzene derivatives.

Scheme 32: Oxidative cyclization of 2,3-diphenylnaphthalenes.

Scheme 33: Suzuki-Miyaura/isomerization/ring closing metathesis strategy to synthesize benz[a]anthracenes.

Scheme 34: Green synthesis of oxa-aza-benzo[a]anthracene and oxa-aza-phenanthrene derivatives.

Scheme 35: Triple benzannulation of substituted naphtalene via a 1,3,6-naphthotriyne synthetic equivalent.

Scheme 36: Zinc iodide-catalyzed Diels–Alder reactions with 1,3-dienes and aroyl propiolates followed by intra...

Scheme 37: H3PO4-promoted intramolecular cyclization of substituted benzoic acids.

Scheme 38: Palladium-catalyzed intermolecular direct acylation of aromatic aldehydes and o-iodoesters.

Scheme 39: Cycloaddition/oxidative aromatization of quinone and β-enamino esters.

Scheme 40: ʟ-Proline-catalyzed [4 + 2] cycloaddition reaction of naphthoquinones and α,β-unsaturated aldehydes....

Scheme 41: Iridium-catalyzed [2 + 2 + 2] cycloaddition of a 1,2-bis(propiolyl)benzene derivative with alkynes.

Scheme 42: Synthesis of several anthraquinone derivatives by using InCl3 and molecular iodine.

Scheme 43: Indium-catalyzed multicomponent reactions employing 2-hydroxy-1,4-naphthoquinone (186), β-naphthol (...

Scheme 44: Synthesis of substituted anthraquinones catalyzed by an AlCl3/MeSO3H system.

Scheme 45: Palladium(II)-catalyzed/visible light-mediated synthesis of anthraquinones.

Scheme 46: [4 + 2] Anionic annulation reaction for the synthesis of substituted anthraquinones.
On the application of 3d metals for C–H activation toward bioactive compounds: The key step for the synthesis of silver bullets
- Renato L. Carvalho,
- Amanda S. de Miranda,
- Mateus P. Nunes,
- Roberto S. Gomes,
- Guilherme A. M. Jardim and
- Eufrânio N. da Silva Júnior
Beilstein J. Org. Chem. 2021, 17, 1849–1938, doi:10.3762/bjoc.17.126

- synthetic methodologies is the C–H bond activation process that enables a straightforward access to several important and innovative compounds [14][15][16][17][18]. In the last few years, metals such as ruthenium [19][20][21], rhodium [22][23][24], palladium [25][26][27], and iridium [28][29][30] have been
- widely applied as catalysts for this matter, including in the synthesis of bioactive substances. Although catalysts based on these metals, are known to be efficient in C–H bond activation reactions affording the products in good yields and mild conditions, they are also known to be usually expensive

Graphical Abstract

Scheme 1: Schematic overview of transition metals studied in C–H activation processes.

Scheme 2: (A) Known biological activities related to benzimidazole-based compounds; (B and C) an example of a...

Scheme 3: (A) Known biological activities related to quinoline-based compounds; (B and C) an example of a sca...

Scheme 4: (A) Known biological activities related to sulfur-containing compounds; (B and C) an example of a s...

Scheme 5: (A) Known biological activities related to aminoindane derivatives; (B and C) an example of a scand...

Scheme 6: (A) Known biological activities related to norbornane derivatives; (B and C) an example of a scandi...

Scheme 7: (A) Known biological activities related to aniline derivatives; (B and C) an example of a titanium-...

Scheme 8: (A) Known biological activities related to cyclohexylamine derivatives; (B) an example of an intram...

Scheme 9: (A) Known biologically active benzophenone derivatives; (B and C) photocatalytic oxidation of benzy...

Scheme 10: (A) Known bioactive fluorine-containing compounds; (B and C) vanadium-mediated C(sp3)–H fluorinatio...

Scheme 11: (A) Known biologically active Lythraceae alkaloids; (B) synthesis of (±)-decinine (30).

Scheme 12: (A) Synthesis of (R)- and (S)-boehmeriasin (31); (B) synthesis of phenanthroindolizidines by vanadi...

Scheme 13: (A) Known bioactive BINOL derivatives; (B and C) vanadium-mediated oxidative coupling of 2-naphthol...

Scheme 14: (A) Known antiplasmodial imidazopyridazines; (B) practical synthesis of 41.

Scheme 15: (A) Gold-catalyzed drug-release mechanism using 2-alkynylbenzamides; (B and C) chromium-mediated al...

Scheme 16: (A) Examples of anti-inflammatory benzaldehyde derivatives; (B and C) chromium-mediated difunctiona...

Scheme 17: (A and B) Manganese-catalyzed chemoselective intramolecular C(sp3)–H amination; (C) late-stage modi...

Scheme 18: (A and B) Manganese-catalyzed C(sp3)–H amination; (C) late-stage modification of a leelamine deriva...

Scheme 19: (A) Known bioactive compounds containing substituted N-heterocycles; (B and C) manganese-catalyzed ...

Scheme 20: (A) Known indoles that present GPR40 full agonist activity; (B and C) manganese-catalyzed C–H alkyl...

Scheme 21: (A) Examples of known biaryl-containing drugs; (B and C) manganese-catalyzed C–H arylation through ...

Scheme 22: (A) Known zidovudine derivatives with potent anti-HIV properties; (B and C) manganese-catalyzed C–H...

Scheme 23: (A and B) Manganese-catalyzed C–H organic photo-electrosynthesis; (C) late-stage modification.

Scheme 24: (A) Example of a known antibacterial silylated dendrimer; (B and C) manganese-catalyzed C–H silylat...

Scheme 25: (A and B) Fe-based small molecule catalyst applied for selective aliphatic C–H oxidations; (C) late...

Scheme 26: (A) Examples of naturally occurring gracilioethers; (B) the first total synthesis of gracilioether ...

Scheme 27: (A and B) Selective aliphatic C–H oxidation of amino acids; (C) late-stage modification of proline-...

Scheme 28: (A) Examples of Illicium sesquiterpenes; (B) first chemical synthesis of (+)-pseudoanisatin (80) in...

Scheme 29: (A and B) Fe-catalyzed deuteration; (C) late-stage modification of pharmaceuticals.

Scheme 30: (A and B) Biomimetic Fe-catalyzed aerobic oxidation of methylarenes to benzaldehydes (PMHS, polymet...

Scheme 31: (A) Known tetrahydroquinolines with potential biological activities; (B and C) redox-selective Fe c...

Scheme 32: (A) Known drugs containing a benzofuran unit; (B and C) Fe/Cu-catalyzed tandem O-arylation to acces...

Scheme 33: (A) Known azaindolines that act as M4 muscarinic acetylcholine receptor agonists; (B and C) intramo...

Scheme 34: (A) Known indolinones with anticholinesterase activity; (B and C) oxidative C(sp3)–H cross coupling...

Scheme 35: (A and B) Cobalt-catalyzed C–H alkenylation of C-3-peptide-containing indoles; (C) derivatization b...

Scheme 36: (A) Cobalt-Cp*-catalyzed C–H methylation of known drugs; (B and C) scope of the o-methylated deriva...

Scheme 37: (A) Known lasalocid A analogues; (B and C) three-component cobalt-catalyzed C–H bond addition; (D) ...

Scheme 38: (A and B) Cobalt-catalyzed C(sp2)–H amidation of thiostrepton.

Scheme 39: (A) Known 4H-benzo[d][1,3]oxazin-4-one derivatives with hypolipidemic activity; (B and C) cobalt-ca...

Scheme 40: (A and B) Cobalt-catalyzed C–H arylation of pyrrole derivatives; (C) application for the synthesis ...

Scheme 41: (A) Known 2-phenoxypyridine derivatives with potent herbicidal activity; (B and C) cobalt-catalyzed...

Scheme 42: (A) Natural cinnamic acid derivatives; (B and C) cobalt-catalyzed C–H carboxylation of terminal alk...

Scheme 43: (A and B) Cobalt-catalyzed C–H borylation; (C) application to the synthesis of flurbiprofen.

Scheme 44: (A) Benzothiazoles known to present anticonvulsant activities; (B and C) cobalt/ruthenium-catalyzed...

Scheme 45: (A and B) Cobalt-catalyzed oxygenation of methylene groups towards ketone synthesis; (C) synthesis ...

Scheme 46: (A) Known anticancer tetralone derivatives; (B and C) cobalt-catalyzed C–H difluoroalkylation of ar...

Scheme 47: (A and B) Cobalt-catalyzed C–H thiolation; (C) application in the synthesis of quetiapine (153).

Scheme 48: (A) Known benzoxazole derivatives with anticancer, antifungal, and antibacterial activities; (B and...

Scheme 49: (A and B) Cobalt-catalyzed C–H carbonylation of naphthylamides; (C) BET inhibitors 158 and 159 tota...

Scheme 50: (A) Known bioactive pyrrolo[1,2-a]quinoxalin-4(5H)-one derivatives; (B and C) cobalt-catalyzed C–H ...

Scheme 51: (A) Known antibacterial cyclic sulfonamides; (B and C) cobalt-catalyzed C–H amination of propargyli...

Scheme 52: (A and B) Cobalt-catalyzed intramolecular 1,5-C(sp3)–H amination; (C) late-stage functionalization ...

Scheme 53: (A and B) Cobalt-catalyzed C–H/C–H cross-coupling between benzamides and oximes; (C) late-state syn...

Scheme 54: (A) Known anticancer natural isoquinoline derivatives; (B and C) cobalt-catalyzed C(sp2)–H annulati...

Scheme 55: (A) Enantioselective intramolecular nickel-catalyzed C–H activation; (B) bioactive obtained motifs;...

Scheme 56: (A and B) Nickel-catalyzed α-C(sp3)–H arylation of ketones; (C) application of the method using kno...

Scheme 57: (A and B) Nickel-catalyzed C(sp3)–H acylation of pyrrolidine derivatives; (C) exploring the use of ...

Scheme 58: (A) Nickel-catalyzed C(sp3)–H arylation of dioxolane; (B) library of products obtained from biologi...

Scheme 59: (A) Intramolecular enantioselective nickel-catalyzed C–H cycloalkylation; (B) product examples, inc...

Scheme 60: (A and B) Nickel-catalyzed C–H deoxy-arylation of azole derivatives; (C) late-stage functionalizati...

Scheme 61: (A and B) Nickel-catalyzed decarbonylative C–H arylation of azole derivatives; (C) application of t...

Scheme 62: (A and B) Another important example of nickel-catalyzed C–H arylation of azole derivatives; (C) app...

Scheme 63: (A and B) Another notable example of a nickel-catalyzed C–H arylation of azole derivatives; (C) lat...

Scheme 64: (A and B) Nickel-based metalorganic framework (MOF-74-Ni)-catalyzed C–H arylation of azole derivati...

Scheme 65: (A) Known commercially available benzothiophene-based drugs; (B and C) nickel-catalyzed C–H arylati...

Scheme 66: (A) Known natural tetrahydrofuran-containing substances; (B and C) nickel-catalyzed photoredox C(sp3...

Scheme 67: (A and B) Another notable example of a nickel-catalyzed photoredox C(sp3)–H alkylation/arylation; (...

Scheme 68: (A) Electrochemical/nickel-catalyzed C–H alkoxylation; (B) achieved scope, including three using na...

Scheme 69: (A) Enantioselective photoredox/nickel catalyzed C(sp3)–H arylation; (B) achieved scope, including ...

Scheme 70: (A) Known commercially available trifluoromethylated drugs; (B and C) nickel-catalyzed C–H trifluor...

Scheme 71: (A and B) Stereoselective nickel-catalyzed C–H difluoroalkylation; (C) late-stage functionalization...

Scheme 72: (A) Cu-mediated ortho-amination of oxalamides; (B) achieved scope, including derivatives obtained f...

Scheme 73: (A) Electro-oxidative copper-mediated amination of 8-aminoquinoline-derived amides; (B) achieved sc...

Scheme 74: (A and B) Cu(I)-mediated C–H amination with oximes; (C) derivatization using telmisartan (241) as s...

Scheme 75: (A and B) Cu-mediated amination of aryl amides using ammonia; (C) late-stage modification of proben...

Scheme 76: (A and B) Synthesis of purine nucleoside analogues using copper-mediated C(sp2)–H activation.

Scheme 77: (A) Copper-mediated annulation of acrylamide; (B) achieved scope, including the synthesis of the co...

Scheme 78: (A) Known bioactive compounds containing a naphthyl aryl ether motif; (B and C) copper-mediated eth...

Scheme 79: (A and B) Cu-mediated alkylation of N-oxide-heteroarenes; (C) late-stage modification.

Scheme 80: (A) Cu-mediated cross-dehydrogenative coupling of polyfluoroarenes and alkanes; (B) scope from know...

Scheme 81: (A) Known anticancer acrylonitrile compounds; (B and C) Copper-mediated cyanation of unactivated al...

Scheme 82: (A) Cu-mediated radiofluorination of 8-aminoquinoline-derived aryl amides; (B) achieved scope, incl...

Scheme 83: (A) Examples of natural β-carbolines; (B and C) an example of a zinc-catalyzed C–H functionalizatio...

Scheme 84: (A) Examples of anticancer α-aminophosphonic acid derivatives; (B and C) an example of a zinc-catal...
A recent overview on the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles
- Pezhman Shiri,
- Ali Mohammad Amani and
- Thomas Mayer-Gall
Beilstein J. Org. Chem. 2021, 17, 1600–1628, doi:10.3762/bjoc.17.114
- . Later, carboxylate-ligand-assisted C–H bond activation takes place through a concerted metalation–deprotonation transformation to produce the next intermediate. Finally, the corresponding product 142 is formed by a reductive elimination process, along with the regeneration of the active catalytic
- '. The isocyanide is inserted into intermediate 158' to achieve intermediate 159'. A 1,2,3-triazole C–H bond activation occurs using palladium, which subsequently undergoes a reductive elimination process to afford final product 154. Likewise, the Pd(0) species is reformed for the next cycle [64]. The

Graphical Abstract

Figure 1: Some significant triazole derivatives [8,23-27].

Scheme 1: A general comparison between synthetic routes for disubstituted 1,2,3-triazole derivatives and full...

Scheme 2: Synthesis of formyltriazoles 3 from the treatment of α-bromoacroleins 1 with azides 2.

Scheme 3: A probable mechanism for the synthesis of formyltriazoles 5 from the treatment of α-bromoacroleins 1...

Scheme 4: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 8 from the reaction of aryl azides 7 with enamino...

Scheme 5: Proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles from the reaction of a...

Scheme 6: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reaction of primary amines 10 with 1,...

Scheme 7: The proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reacti...

Scheme 8: Synthesis of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side chain.

Scheme 9: Mechanism for the formation of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side ch...

Scheme 10: Synthesis of fully decorated 1,2,3-triazole compounds 25 through the regioselective addition and cy...

Scheme 11: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazole compounds 25 through the...

Scheme 12: Synthesis of 1,4,5-trisubstituted glycosyl-containing 1,2,3-triazole derivatives 30 from the reacti...

Scheme 13: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 34 via intramolecular cyclization reaction of ket...

Scheme 14: Synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of aldehydes 35, amines 36, and α...

Scheme 15: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of...

Scheme 16: Synthesis of functionally rich double C- and N-vinylated 1,2,3-triazoles 45 and 47.

Scheme 17: Synthesis of disubstituted 4-chloro-, 4-bromo-, and 4-iodo-1,2,3-triazoles 50.

Scheme 18: a) A general route for SPAAC in polymer chemistry and b) synthesis of a novel pH-sensitive polymeri...

Scheme 19: Synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkynes 57, azides 58, and propargyli...

Scheme 20: A reasonable mechanism for the synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkyne...

Scheme 21: Synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 22: A reasonable mechanism for the synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 23: Synthesis of sulfur-cycle-fused 1,2,3-triazoles 75 and 77.

Scheme 24: A reasonable mechanism for the synthesis of sulfur-cycle-fused 1,2,3‐triazoles 75 and 77.

Scheme 25: Synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, organic azides 83, and ...

Scheme 26: A mechanism for the synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, org...

Scheme 27: Synthesis of trisubstituted triazoles containing an Sb substituent at position C5 in 93 and 5-unsub...

Scheme 28: Synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-butyltosyl disulfide 97...

Scheme 29: A mechanism for the synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-bu...

Scheme 30: Synthesis of triazole-fused sultams 104.

Scheme 31: Synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 32: A reasonable mechanism for the synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 33: Synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 34: A reasonable mechanism for the synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 35: Synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 36: A probable mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 37: Synthesis of fully decorated triazoles 125 via the Pd/C-catalyzed arylation of disubstituted triazo...

Scheme 38: Synthesis of triazolo[1,5-a]indolones 131.

Scheme 39: Synthesis of unsymmetrically substituted triazole-fused enediyne systems 135 and 5-aryl-4-ethynyltr...

Scheme 40: Synthesis of Pd/Cu-BNP 139 and application of 139 in the synthesis of polycyclic triazoles 142.

Scheme 41: A probable mechanism for the synthesis of polycyclic triazoles 142.

Scheme 42: Synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-membered rings 152–154.

Scheme 43: A probable mechanism for the synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-...

Scheme 44: Synthesis of fully functionalized 1,2,3-triazolo-fused chromenes 162, 164, and 166 via the intramol...

Scheme 45: Ru-catalyzed synthesis of fully decorated triazoles 172.

Scheme 46: Synthesis of 4-cyano-1,2,3-triazoles 175.

Scheme 47: Synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 and azides 177 and ...

Scheme 48: Mechanism for the synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 a...
Prins cyclization-mediated stereoselective synthesis of tetrahydropyrans and dihydropyrans: an inspection of twenty years
- Asha Budakoti,
- Pradip Kumar Mondal,
- Prachi Verma and
- Jagadish Khamrai
Beilstein J. Org. Chem. 2021, 17, 932–963, doi:10.3762/bjoc.17.77
- enantioselectivity varied with the polarity of the solvent. The optimization study disclosed that the enantioselectivity increases with the decrease of the polarity of the solvent (Scheme 68). Yu and co-workers reported a novel segment-coupling Prins cyclization involving sequential benzylic/allylic C–H bond
- activation via DDQ oxidation, followed by nucleophilic attack of an unactivated olefin to obtain all-cis-trisubstituted Prins products with high stereochemical precision [111]. A single-electron transfer (SET) mechanism was proposed for the above transformation (Scheme 69). A SET from an arene or alkene to

Graphical Abstract

Scheme 1: General strategy for the synthesis of THPs.

Scheme 2: Developments towards the Prins cyclization.

Scheme 3: General stereochemical outcome of the Prins cyclization.

Scheme 4: Regioselectivity in the Prins cyclization.

Scheme 5: Mechanism of the oxonia-Cope reaction in the Prins cyclization.

Scheme 6: Cyclization of electron-deficient enantioenriched alcohol 27.

Scheme 7: Partial racemization through 2-oxonia-Cope allyl transfer.

Scheme 8: Partial racemization by reversible 2-oxonia-Cope rearrangement.

Scheme 9: Rychnovsky modification of the Prins cyclization.

Scheme 10: Synthesis of (−)-centrolobine and the C22–C26 unit of phorboxazole A.

Scheme 11: Axially selective Prins cyclization by Rychnovsky et al.

Scheme 12: Mechanism for the axially selectivity Prins cyclization.

Scheme 13: Mukaiyama aldol–Prins cyclization reaction.

Scheme 14: Application of the aldol–Prins reaction.

Scheme 15: Hart and Bennet's acid-promoted Prins cyclization.

Scheme 16: Tetrahydropyran core of polycarvernoside A as well as (−)-clavoslide A and D.

Scheme 17: Scheidt and co-workers’ route to tetrahydropyran-4-one.

Scheme 18: Mechanism for the Lewis acid-catalyzed synthesis of tetrahydropyran-4-one.

Scheme 19: Hoveyda and co-workers’ strategy for 2,6-disubstituted 4-methylenetetrahydropyran.

Scheme 20: Funk and Cossey’s ene-carbamates strategy.

Scheme 21: Yadav and Kumar’s cyclopropane strategy for THP synthesis.

Scheme 22: 2-Arylcylopropylmethanolin in centrolobine synthesis.

Scheme 23: Yadav and co-workers’ strategy for the synthesis of THP.

Scheme 24: Yadav and co-workers’ Prins–Ritter reaction sequence for 4-amidotetrahydropyran.

Scheme 25: Yadav and co-workers’ strategy to prelactones B, C, and V.

Scheme 26: Yadav and co-workers’ strategy for the synthesis of (±)-centrolobine.

Scheme 27: Loh and co-workers’ strategy for the synthesis of zampanolide and dactylolide.

Scheme 28: Loh and Chan’s strategy for THP synthesis.

Scheme 29: Prins cyclization of cyclohexanecarboxaldehyde.

Scheme 30: Prins cyclization of methyl ricinoleate (127) and benzaldehyde (88).

Scheme 31: AlCl3-catalyzed cyclization of homoallylic alcohol 129 and aldehyde 130.

Scheme 32: Martín and co-workers’ stereoselective approach for the synthesis of highly substituted tetrahydrop...

Scheme 33: Ene-IMSC strategy by Marko and Leroy for the synthesis of tetrahydropyran.

Scheme 34: Marko and Leroy’s strategy for the synthesis of tetrahydropyrans 146.

Scheme 35: Sakurai dimerization/macrolactonization reaction for the synthesis of cyanolide A.

Scheme 36: Hoye and Hu’s synthesis of (−)-dactyloide by intramolecular Sakurai cyclization.

Scheme 37: Minehan and co-workers’ strategy for the synthesis of THPs 157.

Scheme 38: Yu and co-workers’ allylic transfer strategy for the construction of tetrahydropyran 161.

Scheme 39: Reactivity enhancement in intramolecular Prins cyclization.

Scheme 40: Floreancig and co-workers’ Prins cyclization strategy to (+)-dactyloide.

Scheme 41: Panek and Huang’s DHP synthesis from crotylsilanes: a general strategy.

Scheme 42: Panek and Huang’s DHP synthesis from syn-crotylsilanes.

Scheme 43: Panek and Huang’s DHP synthesis from anti-crotylsilanes.

Scheme 44: Roush and co-workers’ [4 + 2]-annulation strategy for DHP synthesis [82].

Scheme 45: TMSOTf-promoted annulation reaction.

Scheme 46: Dobb and co-workers’ synthesis of DHP.

Scheme 47: BiBr3-promoted tandem silyl-Prins reaction by Hinkle et al.

Scheme 48: Substrate scope of Hinkle and co-workers’ strategy.

Scheme 49: Cho and co-workers’ strategy for 2,6 disubstituted 3,4-dimethylene-THP.

Scheme 50: Furman and co-workers’ THP synthesis from propargylsilane.

Scheme 51: THP synthesis from silyl enol ethers.

Scheme 52: Rychnovsky and co-workers’ strategy for THP synthesis from hydroxy-substituted silyl enol ethers.

Scheme 53: Li and co-workers’ germinal bissilyl Prins cyclization strategy to (−)-exiguolide.

Scheme 54: Xu and co-workers’ hydroiodination strategy for THP.

Scheme 55: Wang and co-workers’ strategy for tetrahydropyran synthesis.

Scheme 56: FeCl3-catalyzed synthesis of DHP from alkynylsilane alcohol.

Scheme 57: Martín, Padrón, and co-workers’ proposed mechanism of alkynylsilane Prins cyclization for the synth...

Scheme 58: Marko and co-workers’ synthesis of 2,6-anti-configured tetrahydropyran.

Scheme 59: Loh and co-workers’ strategy for 2,6-syn-tetrahydropyrans.

Scheme 60: Loh and co-workers’ strategy for anti-THP synthesis.

Scheme 61: Cha and co-workers’ strategy for trans-2,6-tetrahydropyran.

Scheme 62: Mechanism proposed by Cha et al.

Scheme 63: TiCl4-mediated cyclization to trans-THP.

Scheme 64: Feng and co-workers’ FeCl3-catalyzed Prins cyclization strategy to 4-hydroxy-substituted THP.

Scheme 65: Selectivity profile of the Prins cyclization under participation of an iron ligand.

Scheme 66: Sequential reactions involving Prins cyclization.

Scheme 67: Banerjee and co-workers’ strategy of Prins cyclization from cyclopropane carbaldehydes and propargy...

Scheme 68: Mullen and Gagné's (R)-[(tolBINAP)Pt(NC6F5)2][SbF6]2-catalyzed asymmetric Prins cyclization strateg...

Scheme 69: Yu and co-workers’ DDQ-catalyzed asymmetric Prins cyclization strategy to trisubstituted THPs.

Scheme 70: Lalli and Weghe’s chiral-Brønsted-acid- and achiral-Lewis-acid-promoted asymmetric Prins cyclizatio...

Scheme 71: List and co-workers’ iIDP Brønsted acid-promoted asymmetric Prins cyclization strategy.

Scheme 72: Zhou and co-workers’ strategy for chiral phosphoric acid (CPA)-catalyzed cascade Prins cyclization.

Scheme 73: List and co-workers’ approach for asymmetric Prins cyclization using chiral imidodiphosphoric acid ...
Photosensitized direct C–H fluorination and trifluoromethylation in organic synthesis
- Shahboz Yakubov and
- Joshua P. Barham
Beilstein J. Org. Chem. 2020, 16, 2151–2192, doi:10.3762/bjoc.16.183
- catalysis and photochemistry. Where transition metal catalysis provides one strategy for C–H bond activation, transition-metal-free photochemical C–H fluorination can provide a complementary selectivity via a radical mechanism that proceeds under milder conditions than thermal radical activation methods

Graphical Abstract

Figure 1: Fluorine-containing drugs.

Figure 2: Fluorinated agrochemicals.

Scheme 1: Selectivity of fluorination reactions.

Scheme 2: Different mechanisms of photocatalytic activation. Sub = substrate.

Figure 3: Jablonski diagram showing visible-light-induced energy transfer pathways: a) absorption, b) IC, c) ...

Figure 4: Schematic illustration of TTET.

Figure 5: Organic triplet PSCats.

Figure 6: Additional organic triplet PSCats.

Figure 7: A) Further organic triplet PSCats and B) transition metal triplet PSCats.

Figure 8: Different fluorination reagents grouped by generation.

Scheme 3: Synthesis of Selectfluor®.

Scheme 4: General mechanism of PS TTET C(sp3)–H fluorination.

Scheme 5: Selective benzylic mono- and difluorination using 9-fluorenone and xanthone PSCats, respectively.

Scheme 6: Chen’s photosensitized monofluorination: reaction scope.

Scheme 7: Chen’s photosensitized benzylic difluorination reaction scope.

Scheme 8: Photosensitized monofluorination of ethylbenzene on a gram scale.

Scheme 9: Substrate scope of Tan’s AQN-photosensitized C(sp3)–H fluorination.

Scheme 10: AQN-photosensitized C–H fluorination reaction on a gram scale.

Scheme 11: Reaction mechanism of the AQN-assisted fluorination.

Figure 9: 3D structures of the singlet ground and triplet excited states of Selectfluor®.

Scheme 12: Associated transitions for the activation of acetophenone by violet light.

Scheme 13: Ethylbenzene C–H fluorination with various PSCats and conditions.

Scheme 14: Effect of different PSCats on the C(sp3)–H fluorination of cyclohexane (39).

Scheme 15: Reaction scope of Chen’s acetophenone-photosensitized C(sp3)–H fluorination reaction.

Figure 10: a) Site-selectivity of Chen’s acetophenone-photosensitized C–H fluorination reaction [201]. b) Site-sele...

Scheme 16: Formation of the AQN–Selectfluor® exciplex Int1.

Scheme 17: Generation of the C3 2° pentane radical and the Selectfluor® N-radical cation from the exciplex.

Scheme 18: Hydrogen atom abstraction by the Selectfluor® N-radical cation from pentane to give the C3 2° penta...

Scheme 19: Fluorine atom transfer from Selectfluor® to the C3 2° pentane radical to yield 3-fluoropentane and ...

Scheme 20: Barrierless fluorine atom transfer from Int1 to the C3 2° pentane radical to yield 3-fluoropentane,...

Scheme 21: Ketone-directed C(sp3)–H fluorination.

Scheme 22: Ketone-directed fluorination through a 5- and a 6-membered transition state, respectively.

Scheme 23: Effect of different PSCats on the photosensitized C(sp3)–H fluorination of 47.

Scheme 24: Substrate scope of benzil-photoassisted C(sp3)–H fluorinations.

Scheme 25: A) Benzil-photoassisted enone-directed C(sp3)–H fluorination. B) Classification of the reaction mod...

Scheme 26: A) Xanthone-photoassisted ketal-directed C(sp3)–H fluorination. B) Substrate scope. C) C–H fluorina...

Scheme 27: Rationale for the selective HAT at the C2 C–H bond of galactose acetonide.

Scheme 28: Photosensitized C(sp3)–H benzylic fluorination of a peptide using different PSCats.

Scheme 29: Peptide scope of 5-benzosuberenone-photoassisted C(sp3)–H fluorinations.

Scheme 30: Continuous flow PS TTET monofluorination of 72.

Scheme 31: Photosensitized C–H fluorination of N-butylphthalimide as a PSX.

Scheme 32: Substrate scope and limitations of the PSX C(sp3)–H monofluorination.

Scheme 33: Substrate crossover monofluorination experiment.

Scheme 34: PS TTET mechanism proposed by Hamashima and co-workers.

Scheme 35: Photosensitized TFM of 78 to afford α-trifluoromethylated ketone 80.

Scheme 36: Substrate scope for photosensitized styrene TFM to give α-trifluoromethylated ketones.

Scheme 37: Control reactions for photosensitized TFM of styrenes.

Scheme 38: Reaction mechanism for photosensitized TFM of styrenes to afford α-trifluoromethylated ketones.

Scheme 39: Reaction conditions for TFMs to yield the cis- and the trans-product, respectively.

Scheme 40: Substrate scope of trifluoromethylated (E)-styrenes.

Scheme 41: Strategies toward trifluoromethylated (Z)-styrenes.

Scheme 42: Substrate scope of trifluoromethylated (Z)-styrenes.

Scheme 43: Reaction mechanism for photosensitized TFM of styrenes to afford E- or Z-products.
When metal-catalyzed C–H functionalization meets visible-light photocatalysis
- Lucas Guillemard and
- Joanna Wencel-Delord
Beilstein J. Org. Chem. 2020, 16, 1754–1804, doi:10.3762/bjoc.16.147

- last decade particular attention has been focused on two modern fields, C–H bond activation, and visible-light-induced photocatalysis. Couplings through C–H bond activation involve the use of non-prefunctionalized substrates that are directly converted into more complex molecules, without the need of a
- , isolated in moderate to good yields. A possible gram scale-up synthesis was also accomplished. Within the mechanistic cycle, after initial vinylic C–H bond activation and insertion of CO into the C–Pd bond, the acylpalladium intermediate is converted into its conjugated analog under DABCO assistance
- these two activation modes. The advantages of the dual catalytic systems over traditional monocatalyzed approaches is also discussed, thus illustrating their sustainable character. Review Visible-light-mediated photocatalysis as a sustainable reoxidation strategy In the context of traditional C–H bond

Graphical Abstract

Figure 1: Concept of dual synergistic catalysis.

Figure 2: Classification of catalytic systems involving two catalysts.

Figure 3: General mechanism for the dual nickel/photoredox catalytic system.

Figure 4: General mechanisms for C–H activation catalysis involving different reoxidation strategies.

Figure 5: Indole synthesis via dual C–H activation/photoredox catalysis.

Figure 6: Proposed mechanism for the indole synthesis via dual catalysis.

Figure 7: Oxidative Heck reaction on arenes via the dual catalysis.

Figure 8: Proposed mechanism for the Heck reaction on arenes via dual catalysis.

Figure 9: Oxidative Heck reaction on phenols via the dual catalysis.

Figure 10: Proposed mechanism for the Heck reaction on phenols via dual catalysis.

Figure 11: Carbazole synthesis via dual C–H activation/photoredox catalysis.

Figure 12: Proposed mechanism for the carbazole synthesis via dual catalysis.

Figure 13: Carbonylation of enamides via the dual C–H activation/photoredox catalysis.

Figure 14: Proposed mechanism for carbonylation of enamides via dual catalysis.

Figure 15: Annulation of benzamides via the dual C–H activation/photoredox catalysis.

Figure 16: Proposed mechanism for the annulation of benzamides via dual catalysis.

Figure 17: Synthesis of indoles via the dual C–H activation/photoredox catalysis.

Figure 18: Proposed mechanism for the indole synthesis via dual catalysis.

Figure 19: General concept of dual catalysis merging C–H activation and photoredox catalysis.

Figure 20: The first example of dual catalysis merging C–H activation and photoredox catalysis.

Figure 21: Proposed mechanism for the C–H arylation with diazonium salts via dual catalysis.

Figure 22: Dual catalysis merging C–H activation/photoredox using diaryliodonium salts.

Figure 23: Direct arylation via the dual catalytic system reported by Xu.

Figure 24: Direct arylation via dual catalytic system reported by Balaraman.

Figure 25: Direct arylation via dual catalytic system reported by Guo.

Figure 26: C(sp3)–H bond arylation via the dual Pd/photoredox catalytic system.

Figure 27: Acetanilide derivatives acylation via the dual C–H activation/photoredox catalysis.

Figure 28: Proposed mechanism for the C–H acylation with α-ketoacids via dual catalysis.

Figure 29: Acylation of azobenzenes via the dual catalysis C–H activation/photoredox.

Figure 30: C2-acylation of indoles via the dual C–H activation/photoredox catalysis.

Figure 31: Proposed mechanism for the C2-acylation of indoles with aldehydes via dual catalysis.

Figure 32: C2-acylation of indoles via the dual C–H activation/photoredox catalysis.

Figure 33: Perfluoroalkylation of arenes via the dual C–H activation/photoredox catalysis.

Figure 34: Proposed mechanism for perfluoroalkylation of arenes via dual catalysis.

Figure 35: Sulfonylation of 1-naphthylamides via the dual C–H activation/photoredox catalysis.

Figure 36: Proposed mechanism for sulfonylation of 1-naphthylamides via dual catalysis.

Figure 37: meta-C–H Alkylation of arenes via visible-light metallaphotocatalysis.

Figure 38: Alternative procedure for meta-C–H alkylation of arenes via metallaphotocatalysis.

Figure 39: Proposed mechanism for meta-C–H alkylation of arenes via metallaphotocatalysis.

Figure 40: C–H borylation of arenes via visible-light metallaphotocatalysis.

Figure 41: Proposed mechanism for C–H borylation of arenes via visible-light metallaphotocatalysis.

Figure 42: Undirected C–H aryl–aryl cross coupling via dual gold/photoredox catalysis.

Figure 43: Proposed mechanism for the undirected C–H aryl–aryl cross-coupling via dual catalysis.

Figure 44: Undirected C–H arylation of (hetero)arenes via dual manganese/photoredox catalysis.

Figure 45: Proposed mechanism for the undirected arylation of (hetero)arenes via dual catalysis.

Figure 46: Photoinduced C–H arylation of azoles via copper catalysis.

Figure 47: Photo-induced C–H chalcogenation of azoles via copper catalysis.

Figure 48: Decarboxylative C–H adamantylation of azoles via dual cobalt/photoredox catalysis.

Figure 49: Proposed mechanism for the C–H adamantylation of azoles via dual catalysis.

Figure 50: General mechanisms for the “classical” (left) and Cu-free variant (right) Sonogoshira reaction.

Figure 51: First example of a dual palladium/photoredox catalysis for Sonogashira-type couplings.

Figure 52: Arylation of terminal alkynes with diazonium salts via dual gold/photoredox catalysis.

Figure 53: Proposed mechanism for the arylation of terminal alkynes via dual catalysis.

Figure 54: C–H Alkylation of alcohols promoted by H-atom transfer (HAT).

Figure 55: Proposed mechanism for the C–H alkylation of alcohols promoted by HAT.

Figure 56: C(sp3)–H arylation of latent nucleophiles promoted by H-atom transfer.

Figure 57: Proposed mechanism for the C(sp3)–H arylation of latent nucleophiles promoted by HAT.

Figure 58: Direct α-arylation of alcohols promoted by H-atom transfer.

Figure 59: Proposed mechanism for the direct α-arylation of alcohols promoted by HAT.

Figure 60: C–H arylation of amines via dual Ni/photoredox catalysis.

Figure 61: Proposed mechanism for the C–H arylation of amines via dual Ni/photoredox catalysis.

Figure 62: C–H functionalization of nucleophiles via excited ketone/nickel dual catalysis.

Figure 63: Proposed mechanism for the C–H functionalization enabled by excited ketones.

Figure 64: Selective sp3–sp3 cross-coupling promoted by H-atom transfer.

Figure 65: Proposed mechanism for the selective sp3–sp3 cross-coupling promoted by HAT.

Figure 66: Direct C(sp3)–H acylation of amines via dual Ni/photoredox catalysis.

Figure 67: Proposed mechanism for the C–H acylation of amines via dual Ni/photoredox catalysis.

Figure 68: C–H hydroalkylation of internal alkynes via dual Ni/photoredox catalysis.

Figure 69: Proposed mechanism for the C–H hydroalkylation of internal alkynes.

Figure 70: Alternative procedure for the C–H hydroalkylation of ynones, ynoates, and ynamides.

Figure 71: Allylic C(sp3)–H activation via dual Ni/photoredox catalysis.

Figure 72: Proposed mechanism for the allylic C(sp3)–H activation via dual Ni/photoredox catalysis.

Figure 73: Asymmetric allylation of aldehydes via dual Cr/photoredox catalysis.

Figure 74: Proposed mechanism for the asymmetric allylation of aldehydes via dual catalysis.

Figure 75: Aldehyde C–H functionalization promoted by H-atom transfer.

Figure 76: Proposed mechanism for the C–H functionalization of aldehydes promoted by HAT.

Figure 77: Direct C–H arylation of strong aliphatic bonds promoted by HAT.

Figure 78: Proposed mechanism for the C–H arylation of strong aliphatic bonds promoted by HAT.

Figure 79: Direct C–H trifluoromethylation of strong aliphatic bonds promoted by HAT.

Figure 80: Proposed mechanism for the C–H trifluoromethylation of strong aliphatic bonds.
In silico rationalisation of selectivity and reactivity in Pd-catalysed C–H activation reactions
- Liwei Cao,
- Mikhail Kabeshov,
- Steven V. Ley and
- Alexei A. Lapkin
Beilstein J. Org. Chem. 2020, 16, 1465–1475, doi:10.3762/bjoc.16.122
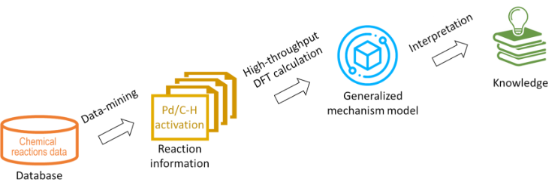
- : https://github.com/sustainable-processes/Pd-catalysed_C-H_activation_reaction_prediction. Results and Discussion Computational approach to rationalise reactivity in Pd-catalysed C–H bond activation reactions Chemical reactivity is simultaneously influenced by many factors including catalysts, reactants

Graphical Abstract

Figure 1: An approximate energy map for the electrophilic aromatic substitution mechanism.

Scheme 1: Schematic representation of the two mechanisms of Pd-catalysed C–H activation reaction considered i...
Synthesis of esters of diaminotruxillic bis-amino acids by Pd-mediated photocycloaddition of analogs of the Kaede protein chromophore
- Esteban P. Urriolabeitia,
- Pablo Sánchez,
- Alexandra Pop,
- Cristian Silvestru,
- Eduardo Laga,
- Ana I. Jiménez and
- Carlos Cativiela
Beilstein J. Org. Chem. 2020, 16, 1111–1123, doi:10.3762/bjoc.16.98
- literature [52][53][54][55][56][57][58][59]. The styryl fragment bonded to an oxazolone ring has not been characterized previously by X-ray methods, but it shows internal bond distances and angles similar to those found in related 2-styrylimidazolone species [60]. C–H Bond activation processes on (Z)-4
- ], did not produce the expected cyclobutanes or gave only very low conversions (<5%) after long reaction times (96 h). As this reactivity was poor, it was decided to attempt the reaction using Pd complexes as templates [28][29][30]. The first step in this process was to study the C–H bond activation in
- factors, namely the presence of bulky CF3 and NO2 groups in ortho positions of the starting oxazolones 2e and 2i, and the strongly deactivating effect of the two groups on the C–H bond activation process. The reactions were usually carried out using 0.6 mmol of the starting material 2. In this respect, it

Graphical Abstract

Figure 1: (a) General scheme for truxillic acid derivatives; (b) general scheme for symmetric 1,3-diaminotrux...

Figure 2: (a) (Z)-4-Arylidene-2-aryl-5(4H)-oxazolones used for the synthesis of 1,3-diaminotruxillic derivati...

Figure 3: (Z)-4-Arylidene-2((E)-styryl)-5(4H)-oxazolones 2a–j used in this work and overall reaction scheme.

Figure 4: Molecular drawing of the oxazolone 2c.

Scheme 1: Ortho-palladation of oxazolones 2 by treatment with Pd(OAc)2 and different structures obtained for ...

Scheme 2: [2 + 2] Photocycloaddition of cyclopalladated complexes 3 in solution to give the dinuclear cyclobu...

Figure 5: Molecular drawing of cyclobutane ortho-palladated 4a. Ellipsoids are shown at the 50% probability l...

Scheme 3: Release of the 1,3-diaminotruxillic bis-amino ester derivatives 5 by methoxycarbonylation of the Pd...
Bipyrrole boomerangs via Pd-mediated tandem cyclization–oxygenation. Controlling reaction selectivity and electronic properties
- Liliia Moshniaha,
- Marika Żyła-Karwowska,
- Joanna Cybińska,
- Piotr J. Chmielewski,
- Ludovic Favereau and
- Marcin Stępień
Beilstein J. Org. Chem. 2020, 16, 895–903, doi:10.3762/bjoc.16.81

- measurements show that helicene-like boomerangs may be usable as circularly polarized luminescent materials. Keywords: donor–acceptor systems; double C–H bond activation; helicenes; pyrroles; Introduction Nanographenes and other polycyclic aromatics as well as their heterocyclic analogues are typically
- disadvantage may be obviated by transition-metal-mediated double C–H bond activation [22][23], which is functionally equivalent to conventional oxidative coupling reactions, and has become a powerful synthetic tool with a rapidly growing scope of use [24][25][26]. However, in the field of π-conjugated
- (NDA) and naphthalenemonoimide (NMI) moieties. The double C–H bond activation initially used palladium(II) acetate in acetic acid as the coupling system. The subsequent screening revealed, however, that a catalytic coupling could be also achieved in the presence of silver(I) carbonate as the

Graphical Abstract

Scheme 1: The previously reported family of the boomerang bipyrroles obtained by Pd-induced double C–H bond a...

Scheme 2: Synthesis and structures of α-free and α-oxygenated bipyrrole boomerangs. Reagents and conditions: ...

Figure 1: DFT-Optimized structures (B3LYP/6-31G(d,p)) of cNDA2O and cNMI3H.

Figure 2: Absorption and emission spectra of cNMI2H (top) and cNMI3H (bottom) measured in toluene, dichlorome...
Synthesis of C70-fragment buckybowls bearing alkoxy substituents
- Yumi Yakiyama,
- Shota Hishikawa and
- Hidehiro Sakurai
Beilstein J. Org. Chem. 2020, 16, 681–690, doi:10.3762/bjoc.16.66

- equilibrium between the Pd(IV) intermediates through C–H bond activation. Keywords: buckybowl; C70; rearrangement through C–H bond activation; Introduction The study of buckybowls, the bowl-shaped π-conjugated aromatic hydrocarbons corresponding to the fragments of fullerenes, pioneered by the works on
- succeeded in synthesizing three different alkoxy-substituted C70-fragment buckybowls 5a–c. In particular, 5c was not an intended molecule, but was formed unexpectedly through the rearrangement through the Pd-catalyzed C–H bond activation reaction. The X-ray crystal structure analysis of 5a–c clearly

Graphical Abstract

Figure 1: Structure of the target buckybowls 5a–c.

Scheme 1: Synthesis of dialkoxides 5a–c.

Scheme 2: Proposed mechanism of the formation of 5b and 5c.

Figure 2: Crystal structure of 5a. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 3: a) Definition of POAV angle (φ). b) Side and c) top view of the molecular skeleton of 1. The double...

Figure 4: Crystal structure of 5b. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 5: Crystal structure of 5c. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 6: a) UV–vis spectra and b) emission spectra of 1 and dialkoxides 5a–c. For all the spectra, the conce...
Architecture and synthesis of P,N-heterocyclic phosphine ligands
- Wisdom A. Munzeiwa,
- Bernard Omondi and
- Vincent O. Nyamori
Beilstein J. Org. Chem. 2020, 16, 362–383, doi:10.3762/bjoc.16.35

- from α-C–H bond activation in heterocycles The α-position to a heteroatom in a cyclic compound is activated because of the difference in electronegativity with carbon. This presents an opportunity to readily generate organometallic nucleophiles. Chelucci et al. [71] used this fact to synthesize the

Graphical Abstract

Scheme 1: Synthesis of pyridylphosphine ligands.

Figure 1: Pyridylphosphine ligands.

Scheme 2: Synthesis of piperidyl- and oxazinylphosphine ligands.

Scheme 3: Synthesis of linear multi-chelate pyridylphosphine ligands.

Scheme 4: Synthesis of chiral acetal pyridylphosphine ligands.

Scheme 5: Synthesis of diphenylphosphine-substituted triazine ligands.

Scheme 6: Synthesis of (pyridine-2-ylmethyl)phosphine ligands.

Scheme 7: Synthesis of diphosphine pyrrole ligands.

Scheme 8: Synthesis of 4,5-diazafluorenylphosphine ligands.

Scheme 9: Synthesis of thioether-containing pyridyldiphosphine ligands starting from ethylene sulfide and dip...

Scheme 10: Synthesis of monoterpene-derived phosphine pyridine ligands.

Scheme 11: Synthesis of N-phenylphosphine-substituted imidazole ligands.

Scheme 12: Synthesis of triazol-4-ylphosphine ligands.

Scheme 13: Synthesis of phosphanyltriazolopyridines and product selectivity depending on the substituents’ eff...

Scheme 14: Synthesis of PTA-phosphine ligands.

Scheme 15: Synthesis of isomeric phosphine dipyrazole ligands by varying the reaction temperature.

Scheme 16: Synthesis of N-tethered phosphine imidazolium ligands (route A) and diphosphine imidazolium ligands...

Scheme 17: Synthesis of {1-[2-(pyridin-2-yl)- (R = CH) and {1-[2-(pyrazin-2-yl)quinazolin-4-yl]naphthalen-2-yl...

Scheme 18: Synthesis of oxazolylindolylphosphine ligands 102.

Scheme 19: Synthesis of pyrrolylphosphine ligands.

Scheme 20: Synthesis of phosphine guanidinium ligands.

Scheme 21: Synthesis of a polydentate aminophosphine ligand.

Scheme 22: Synthesis of quinolylphosphine ligands.

Scheme 23: Synthesis of N-(triazolylmethyl)phosphanamine ligands.

Figure 2: Triazolylphosphanamine ligands synthesized by Wassenaar’s method [22].

Scheme 24: Synthesis of oxazaphosphorines.

Scheme 25: Synthesis of paracyclophane pyridylphosphine ligands.

Scheme 26: Synthesis of triazolylphosphine ligands.

Figure 3: Click-phosphine ligands.

Scheme 27: Ferrocenyl pyridylphosphine imine ligands.

Scheme 28: Synthesis of phosphinooxazolines (PHOX).

Scheme 29: Synthesis of ferrocenylphosphine oxazoles.
Recent advances in transition-metal-catalyzed incorporation of fluorine-containing groups
- Xiaowei Li,
- Xiaolin Shi,
- Xiangqian Li and
- Dayong Shi
Beilstein J. Org. Chem. 2019, 15, 2213–2270, doi:10.3762/bjoc.15.218
- intermediate B through the C–H bond-activation process. Oxidative addition of the intermediate B with Selectfluor affords the palladium(IV) species C, followed by reductive elimination and ligand dissociation to give the final product. Similar to these publications in strategy and products, in the same year
- process, an in situ-generated cationic [Pd(NO3)]+ species facilitates the C–H bond activation to give intermediate A. The Pd(II)(1a)2 complex B is formed via further C–H bond activation of another molecule 1a by the cyclopalladation(II) intermediate A. Then, intermediate B undergoes oxidative addition by

Graphical Abstract

Scheme 1: The main three strategies of fluorination: nucleophilic, electrophilic and radical fluorination.

Scheme 2: Doyle’s Pd-catalyzed fluorination of allylic chlorides.

Scheme 3: Allylic fluorination of 2- and 3-substituted propenyl esters.

Scheme 4: Regioselective allylic fluorination of cinnamyl phosphorothioate esters.

Scheme 5: Palladium-catalyzed aliphatic C–H fluorination reported by Doyle.

Scheme 6: Pd-catalyzed enantioselective fluorination of α-ketoesters followed by stereoselective reduction to...

Scheme 7: Pd-catalyzed C(sp3)–H fluorination of oxindoles.

Scheme 8: C–H fluorination of 8-methylquinoline derivatives with F− reagents.

Scheme 9: Fluorination of α-cyano acetates reported by van Leeuwen.

Scheme 10: The catalytic enantioselective electrophilic C–H fluorination of α-chloro-β-keto phosphonates.

Scheme 11: Fluorination of unactivated C(sp3)–H bonds directed by the bidentate PIP auxiliary.

Scheme 12: Fluorination of C(sp3)–H bonds at the β-position of carboxylic acids.

Scheme 13: Enantioselective benzylic C–H fluorination with a chiral transient directing group.

Scheme 14: Microwave-heated Pd-catalyzed fluorination of aryl alcohols.

Scheme 15: Fluorination of aryl potassium trifluoroborates.

Scheme 16: C(sp2)–F bond formation using precatalyst [L·Pd]2(cod).

Scheme 17: Pd-catalyzed fluorination of (hetero)aryl triflates and bromides.

Scheme 18: The Pd-catalyzed C–H fluorination of arenes with Selectfluor/NFSI.

Scheme 19: Pd(II)-catalyzed ortho-monofluorination protocol for benzoic acids.

Scheme 20: Pd-catalyzed C(sp2)–H bond fluorination of 2-arylbenzothiazoles.

Scheme 21: Nitrate-promoted fluorination of aromatic and olefinic C(sp2)–H bonds and proposed mechanism.

Scheme 22: Fluorination of oxalyl amide-protected benzylamine derivatives.

Scheme 23: C–H fluorination of benzaldehydes with orthanilic acids as transient directing group.

Scheme 24: Pd(II)-catalyzed aryl C–H fluorination with various directing groups.

Scheme 25: Cu-catalyzed aliphatic, allylic, and benzylic fluorination.

Scheme 26: Cu-catalyzed SN2 fluorination of primary and secondary alkyl bromides.

Scheme 27: Copper-catalyzed fluorination of alkyl triflates.

Scheme 28: Cu-catalyzed fluorination of allylic bromides and chlorides.

Scheme 29: Synthetic strategy for the fluorination of active methylene compounds.

Scheme 30: Fluorination of β-ketoesters using a tartrate-derived bidentate bisoxazoline-Cu(II) complex.

Scheme 31: Highly enantioselective fluorination of β-ketoesters and N-Boc-oxindoles.

Scheme 32: Amide group-assisted site-selective fluorination of α-bromocarbonyl compounds.

Scheme 33: Cu-mediated aryl fluorination reported by Sanford [77].

Scheme 34: Mono- or difluorination reactions of benzoic acid derivatives.

Scheme 35: Cu-catalyzed fluorination of diaryliodonium salts with KF.

Scheme 36: Copper(I)-catalyzed cross-coupling of 2-pyridylaryl bromides.

Scheme 37: AgNO3-catalyzed decarboxylative fluorination of aliphatic carboxylic acids.

Scheme 38: The Mn-catalyzed aliphatic and benzylic C–H fluorination.

Scheme 39: Iron(II)-promoted C–H fluorination of benzylic substrates.

Scheme 40: Ag-catalyzed fluorodecarboxylation of carboxylic acids.

Scheme 41: Vanadium-catalyzed C(sp3)–H fluorination.

Scheme 42: AgNO3-catalyzed radical deboronofluorination of alkylboronates and boronic acids.

Scheme 43: Selective heterobenzylic C–H fluorination with Selectfluor reported by Van Humbeck.

Scheme 44: Fe(II)-catalyzed site-selective fluorination guided by an alkoxyl radical.

Scheme 45: Fluorination of allylic trichloroacetimidates reported by Nguyen et al.

Scheme 46: Iridium-catalyzed fluorination of allylic carbonates with TBAF(t-BuOH)4.

Scheme 47: Iridium-catalyzed asymmetric fluorination of allylic trichloroacetimidates.

Scheme 48: Cobalt-catalyzed α-fluorination of β-ketoesters.

Scheme 49: Nickel-catalyzed α-fluorination of various α-chloro-β-ketoesters.

Scheme 50: Ni(II)-catalyzed enantioselective fluorination of oxindoles and β-ketoesters.

Scheme 51: Scandium(III)-catalyzed asymmetric C–H fluorination of unprotected 3-substituted oxindoles.

Scheme 52: Iron-catalyzed directed C–H fluorination.

Scheme 53: Electrophilic silver-catalyzed Ar–F bond-forming reaction from arylstannanes.

Figure 1: Nucleophilic, electrophilic and radical CF3 sources.

Scheme 54: Cu(I)-catalyzed allylic trifluoromethylation of unactivated terminal olefins.

Scheme 55: Direct copper-catalyzed trifluoromethylation of allylsilanes.

Scheme 56: Cupper-catalyzed enantioselective trifluoromethylation of five and six-membered ring β-ketoesters.

Scheme 57: Cu-catalyzed highly stereoselective trifluoromethylation of secondary propargyl sulfonates.

Scheme 58: Remote C(sp3)–H trifluoromethylation of carboxamides and sulfonamides.

Scheme 59: Trifluoromethylation of allylsilanes with photoredox catalysis.

Scheme 60: Ag-catalyzed decarboxylative trifluoromethylation of aliphatic carboxylic acids in aqueous CH3CN.

Scheme 61: Decarboxylative trifluoromethylation of aliphatic carboxylic acids via combined photoredox and copp...

Scheme 62: Palladium-catalyzed Ar–CF3 bond-forming reaction.

Scheme 63: Palladium-catalyzed trifluoromethylation of arenes with diverse heterocyclic directing groups.

Scheme 64: Pd-catalyzed trifluoromethylation of indoles as reported by Liu.

Scheme 65: Pd-catalyzed trifluoromethylation of vinyl triflates and vinyl nonaflates.

Scheme 66: Pd(II)-catalyzed ortho-trifluoromethylation of aromatic C–H bonds.

Scheme 67: Visible-light-induced Pd(OAc)2-catalyzed ortho-trifluoromethylation of acetanilides with CF3SO2Na.

Scheme 68: CuI-catalyzed trifluoromethylation of aryl- and alkenylboronic acids.

Scheme 69: Cu-catalyzed trifluoromethylation of aryl- and vinylboronic acids.

Scheme 70: Copper-catalyzed trifluoromethylation of α,β-unsaturated carboxylic acids.

Scheme 71: Formation of C(sp2)–CF3 bond catalyzed by copper(I) complex.

Scheme 72: Loh’s Cu(I)-catalyzed trifluoromethylation of enamides and electron-deficient alkenes.

Scheme 73: Copper and iron-catalyzed decarboxylative tri- and difluoromethylation.

Scheme 74: Cu-catalyzed trifluoromethylation of hydrazones developed by Bouyssi.

Scheme 75: Cu(I)-catalyzed trifluoromethylation of terminal alkenes.

Scheme 76: Cu/Ag-catalyzed decarboxylative trifluoromethylation of cinnamic acids.

Scheme 77: Copper-catalyzed direct alkenyl C–H trifluoromethylation.

Scheme 78: Copper(I/II)-catalyzed direct trifluoromethylation of styrene derivatives.

Scheme 79: Regioselective trifluoromethylation of pivalamido arenes and heteroarenes.

Scheme 80: Synthesis of trifluoromethylquinones in the presence of copper(I).

Scheme 81: Oxidative trifluoromethylation of imidazoheterocycles in ionic liquid/water.

Scheme 82: A mild and fast continuous-flow trifluoromethylation of coumarins using a CuI/CF3SO2Na/TBHP system.

Scheme 83: Copper-catalyzed oxidative trifluoromethylation of various 8-aminoquinolines.

Scheme 84: PA-directed copper-catalyzed trifluoromethylation of anilines.

Scheme 85: Trifluoromethylation of potassium vinyltrifluoroborates catalyzed by Fe(II).

Scheme 86: Alkenyl trifluoromethylation catalyzed by Ru(phen)3Cl2 as photocatalyst.

Scheme 87: Ru-catalyzed trifluoromethylation of alkenes by Akita’s group.

Scheme 88: Ir-catalyzed Cvinyl–CF3 bond formation of α,β-unsaturated carboxylic acids.

Scheme 89: Ag(I)-catalyzed denitrative trifluoromethylation of β-nitrostyrenes.

Scheme 90: Photocatalyzed direct trifluoromethylation of aryl and heteroaryl C–H bonds.

Scheme 91: Rhenium (MTO)-catalyzed direct trifluoromethylation of aromatic substrates.

Scheme 92: Trifluoromethylation of unprotected anilines under [Ir(ppy)3] catalyst.

Scheme 93: Oxidative trifluoromethylation of imidazopyridines and imidazoheterocycles.

Scheme 94: Ruthenium-catalyzed trifluoromethylation of (hetero)arenes with trifluoroacetic anhydride.

Scheme 95: Phosphovanadomolybdic acid-catalyzed direct C–H trifluoromethylation.

Scheme 96: Picolinamide-assisted ortho-trifluoromethylation of arylamines.

Scheme 97: A nickel-catalyzed C–H trifluoromethylation of free anilines.

Scheme 98: Cu-mediated trifluoromethylation of terminal alkynes reported by Qing.

Scheme 99: Huang’s C(sp)–H trifluoromethylation using Togni’s reagent.

Scheme 100: Cu-catalyzed methods for trifluoromethylation with Umemoto’s reagent.

Scheme 101: The synthesis of alkynyl-CF3 compounds in the presence of fac-[Ir(ppy)3] under visible-light irradi...

Scheme 102: Pd-catalyzed Heck reaction reported by Reutrakul.

Scheme 103: Difluoromethylation of enamides and ene-carbamates.

Scheme 104: Difluoromethylation of α,β-unsaturated carboxylic acids.

Scheme 105: Copper-catalyzed direct C(sp2)–H difluoroacetylation reported by Pannecoucke and co-workers.

Scheme 106: Difluoroalkylation of aldehyde-derived hydrazones with functionalized difluoromethyl bromides.

Scheme 107: Photoredox-catalyzed C–H difluoroalkylation of aldehyde-derived hydrazones.

Scheme 108: Synergistic ruthenium(II)-catalyzed C–H difluoromethylation reported by Ackermann.

Scheme 109: Visible-light photocatalytic decarboxylation of α,β-unsaturated carboxylic acids.

Scheme 110: Synthesis of difluorinated ketones via S-alkyl dithiocarbamates obtained from acyl chlorides and po...

Scheme 111: Synthesis of aryl and heteroaryl difluoromethylated phosphonates.

Scheme 112: Difluoroalkylation of secondary propargyl sulfonates using Cu as the catalyst.

Scheme 113: Ru(II)-mediated para-selective difluoromethylation of anilides and their derivatives.

Scheme 114: Bulky diamine ligand promoted cross-coupling of difluoroalkyl bromides.

Scheme 115: Copper-catalyzed C3–H difluoroacetylation of quinoxalinones.

Scheme 116: Copper(I) chloride-catalyzed trifluoromethylthiolation of enamines, indoles and β-ketoesters.

Scheme 117: Copper-boxmi-catalyzed asymmetric trifluoromethylthiolation of β-ketoesters.

Scheme 118: Direct Cu-catalyzed trifluoromethylthiolation of boronic acids and alkynes.

Scheme 119: Cu-catalyzed synthesis of α-trifluoromethylthio-substituted ketones.

Scheme 120: Trifluoromethylthiolation reactions promoted by diazotriflone and copper.

Scheme 121: Halide activation of N-(trifluoromethylthio)phthalimide.

Scheme 122: The visible light-promoted trifluoromethylthiolation reported by Glorius.

Scheme 123: Synthesis of α-trifluoromethylthioesters via Goossen’s approach.

Scheme 124: Photoinduced trifluoromethylthiolation of diazonium salts.

Scheme 125: Ag-mediated trifluoromethoxylation of aryl stannanes and arylboronic acids.

Scheme 126: Catalytic (hetero)aryl C–H trifluoromethoxylation under visible light.

Scheme 127: Photoinduced C–H-bond trifluromethoxylation of (hetero)arenes.
Regioselective Pd-catalyzed direct C1- and C2-arylations of lilolidine for the access to 5,6-dihydropyrrolo[3,2,1-ij]quinoline derivatives
- Hai-Yun Huang,
- Haoran Li,
- Thierry Roisnel,
- Jean-François Soulé and
- Henri Doucet
Beilstein J. Org. Chem. 2019, 15, 2069–2075, doi:10.3762/bjoc.15.204

- methodology provides a straightforward access to several lilolidine derivatives from commercially available compounds via one, two or three C–H bond functionalization steps allowing to tune their biological properties. Keywords: catalysis: C–C bond formation; C–H bond activation; lilolidine; palladium

Graphical Abstract

Figure 1: Structures of lilolidine, tivantinib and tarazepide.

Scheme 1: Access to α- and β-arylated lilolidine derivatives.

Scheme 2: Synthesis of α-arylated lilolidine derivatives.

Scheme 3: Synthesis of α,β-di(hetero)arylated lilolidine derivatives.

Scheme 4: Synthesis of α,β-diarylated lilolidine derivatives via successive direct arylations.

Scheme 5: Synthesis of 5,6-dihydrodibenzo[a,c]pyrido[3,2,1-jk]carbazoles via successive direct arylations.
Attempted synthesis of a meta-metalated calix[4]arene
- Christopher D. Jurisch and
- Gareth E. Arnott
Beilstein J. Org. Chem. 2019, 15, 1996–2002, doi:10.3762/bjoc.15.195
- ) and rhodium(III) complexes underwent spontaneous, chemoselective cyclometalation via C–H bond activation of the N-bound phenyl group (see Figure 2) [19]. Incorporation of a mesoionic carbene directing group onto the upper rim of a calix[4]arene therefore offered potential for a new route to calix[4

Graphical Abstract

Figure 1: Inherent chirality generated by meta-substitution – the two structures are non-superposable mirror ...

Figure 2: General approach by Albrecht for MIC directed cyclometalation via C–H activation; M = Ru(II), Ir(II...

Figure 3: Concept of cyclometalated calix[4]arene target.

Scheme 1: Synthesis of model mesoionic carbene 5.

Scheme 2: Attempted Ullmann-coupling to give monoazide 7.

Scheme 3: Synthesis of monoazidocalix[4]arene 7 under optimized conditions.

Scheme 4: Synthesis of the putative calix[4]arene mesoionic carbene ruthenium complex 13.

Figure 4: High-resolution mass spectrum (ESI+) of putative ruthenacycle calix[4]arene 13.
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
- Gagandeep Kour Reen,
- Ashok Kumar and
- Pratibha Sharma
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165

- high level of functional group compatibility, intramolecular alkylations, inter and intramolecular C–H bond activation and regio/stereoselective homocoupling of alkynes [71][72][73]. Group of Cho and Chang have unprecedentedly reported a Rh catalytic system for facile addition of heteroarenes to both
- ). However, the reaction was unsuccessful with aliphatic and low-boiling aldehydes (as the reaction was taking place at high temperature). The reaction mechanism involved a Cu-NPs-mediated C–H bond activation of the alkyne which further reacted with the iminium ion to form propargylamine 45 and Cu NPs were

Graphical Abstract

Figure 1: Various drugs having IP nucleus.

Figure 2: Participation percentage of various TMs for the syntheses of IPs.

Scheme 1: CuI–NaHSO4·SiO2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 2: Experimental examination of reaction conditions.

Scheme 3: One-pot tandem reaction for the synthesis of 2-haloimidazopyridines.

Scheme 4: Mechanistic scheme for the synthesis of 2-haloimidazopyridine.

Scheme 5: Copper-MOF-catalyzed three-component reaction (3-CR) for imidazo[1,2-a]pyridines.

Scheme 6: Mechanism for copper-MOF-driven synthesis.

Scheme 7: Heterogeneous synthesis via titania-supported CuCl2.

Scheme 8: Mechanism involving oxidative C–H functionalization.

Scheme 9: Heterogeneous synthesis of IPs.

Scheme 10: One-pot regiospecific synthesis of imidazo[1,2-a]pyridines.

Scheme 11: Vinyl azide as an unprecedented substrate for imidazo[1,2-a]pyridines.

Scheme 12: Radical pathway.

Scheme 13: Cu(I)-catalyzed transannulation approach for imidazo[1,5-a]pyridines.

Scheme 14: Plausible radical pathway for the synthesis of imidazo[1,5-a]pyridines.

Scheme 15: A solvent-free domino reaction for imidazo[1,2-a]pyridines.

Scheme 16: Cu-NPs-mediated synthesis of imidazo[1,2-a]pyridines.

Scheme 17: CuI-catalyzed synthesis of isoxazolylimidazo[1,2-a]pyridines.

Scheme 18: Functionalization of 4-bromo derivative via Sonogashira coupling reaction.

Scheme 19: A plausible reaction pathway.

Scheme 20: Cu(I)-catalyzed intramolecular oxidative C–H amidation reaction.

Scheme 21: One-pot synthetic reaction for imidazo[1,2-a]pyridine.

Scheme 22: Plausible reaction mechanism.

Scheme 23: Cu(OAc)2-promoted synthesis of imidazo[1,2-a]pyridines.

Scheme 24: Mechanism for aminomethylation/cycloisomerization of propiolates with imines.

Scheme 25: Three-component synthesis of imidazo[1,2-a]pyridines.

Figure 3: Scope of pyridin-2(1H)-ones and acetophenones.

Scheme 26: CuO NPS-promoted A3 coupling reaction.

Scheme 27: Cu(II)-catalyzed C–N bond formation reaction.

Scheme 28: Mechanism involving Chan–Lam/Ullmann coupling.

Scheme 29: Synthesis of formyl-substituted imidazo[1,2-a]pyridines.

Scheme 30: A tandem sp3 C–H amination reaction.

Scheme 31: Probable mechanistic approach.

Scheme 32: Dual catalytic system for imidazo[1,2-a]pyridines.

Scheme 33: Tentative mechanism.

Scheme 34: CuO/CuAl2O4/ᴅ-glucose-promoted 3-CCR.

Scheme 35: A tandem CuOx/OMS-2-based synthetic strategy.

Figure 4: Biomimetic catalytic oxidation in the presence of electron-transfer mediators (ETMs).

Scheme 36: Control experiment.

Scheme 37: Copper-catalyzed C(sp3)–H aminatin reaction.

Scheme 38: Reaction of secondary amines.

Scheme 39: Probable mechanistic pathway.

Scheme 40: Coupling reaction of α-azidoketones.

Scheme 41: Probable pathway.

Scheme 42: Probable mechanism with free energy calculations.

Scheme 43: MCR for cyanated IP synthesis.

Scheme 44: Substrate scope for the reaction.

Scheme 45: Reaction mechanism.

Scheme 46: Probable mechanistic pathway for Cu/ZnAl2O4-catalyzed reaction.

Scheme 47: Copper-catalyzed double oxidative C–H amination reaction.

Scheme 48: Application towards different coupling reactions.

Scheme 49: Reaction mechanism.

Scheme 50: Condensation–cyclization approach for the synthesis of 1,3-diarylated imidazo[1,5-a]pyridines.

Scheme 51: Optimized reaction conditions.

Scheme 52: One-pot 2-CR.

Scheme 53: One-pot 3-CR without the isolation of chalcone.

Scheme 54: Copper–Pybox-catalyzed cyclization reaction.

Scheme 55: Mechanistic pathway catalyzed by Cu–Pybox complex.

Scheme 56: Cu(II)-promoted C(sp3)-H amination reaction.

Scheme 57: Wider substrate applicability for the reaction.

Scheme 58: Plausible reaction mechanism.

Scheme 59: CuI assisted C–N cross-coupling reaction.

Scheme 60: Probable reaction mechanism involving sp3 C–H amination.

Scheme 61: One-pot MCR-catalyzed by CoFe2O4/CNT-Cu.

Scheme 62: Mechanistic pathway.

Scheme 63: Synthetic scheme for 3-nitroimidazo[1,2-a]pyridines.

Scheme 64: Plausible mechanism for CuBr-catalyzed reaction.

Scheme 65: Regioselective synthesis of halo-substituted imidazo[1,2-a]pyridines.

Scheme 66: Synthesis of 2-phenylimidazo[1,2-a]pyridines.

Scheme 67: Synthesis of diarylated compounds.

Scheme 68: CuBr2-mediated one-pot two-component oxidative coupling reaction.

Scheme 69: Decarboxylative cyclization route to synthesize 1,3-diarylimidazo[1,5-a]pyridines.

Scheme 70: Mechanistic pathway.

Scheme 71: C–H functionalization reaction of enamines to produce diversified heterocycles.

Scheme 72: A plausible mechanism.

Scheme 73: CuI-promoted aerobic oxidative cyclization reaction of ketoxime acetates and pyridines.

Scheme 74: CuI-catalyzed pathway for the formation of imidazo[1,2-a]pyridine.

Scheme 75: Mechanistic pathway.

Scheme 76: Mechanistic rationale for the synthesis of products.

Scheme 77: Copper-catalyzed synthesis of vinyloxy-IP.

Scheme 78: Regioselective product formation with propiolates.

Scheme 79: Proposed mechanism for vinyloxy-IP formation.

Scheme 80: Regioselective synthesis of 3-hetero-substituted imidazo[1,2-a]pyridines with different reaction su...

Scheme 81: Mechanistic pathway.

Scheme 82: CuI-mediated synthesis of 3-formylimidazo[1,2-a]pyridines.

Scheme 83: Radical pathway for 3-formylated IP synthesis.

Scheme 84: Pd-catalyzed urea-cyclization reaction for IPs.

Scheme 85: Pd-catalyzed one-pot-tandem amination and intramolecular amidation reaction.

Figure 5: Scope of aniline nucleophiles.

Scheme 86: Pd–Cu-catalyzed Sonogashira coupling reaction.

Scheme 87: One-pot amide coupling reaction for the synthesis of imidazo[4,5-b]pyridines.

Scheme 88: Urea cyclization reaction for the synthesis of two series of pyridines.

Scheme 89: Amidation reaction for the synthesis of imidazo[4,5-b]pyridines.

Figure 6: Amide scope.

Scheme 90: Pd NPs-catalyzed 3-component reaction for the synthesis of 2,3-diarylated IPs.

Scheme 91: Plausible mechanistic pathway for Pd NPs-catalyzed MCR.

Scheme 92: Synthesis of chromenoannulated imidazo[1,2-a]pyridines.

Scheme 93: Mechanism for the synthesis of chromeno-annulated IPs.

Scheme 94: Zinc oxide NRs-catalyzed synthesis of imidazo[1,2-a]azines/diazines.

Scheme 95: Zinc oxide-catalyzed isocyanide based GBB reaction.

Scheme 96: Reaction pathway for ZnO-catalyzed GBB reaction.

Scheme 97: Mechanistic pathway.

Scheme 98: ZnO NRs-catalyzed MCR for the synthesis of imidazo[1,2-a]azines.

Scheme 99: Ugi type GBB three-component reaction.

Scheme 100: Magnetic NPs-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 101: Regioselective synthesis of 2-alkoxyimidazo[1,2-a]pyridines catalyzed by Fe-SBA-15.

Scheme 102: Plausible mechanistic pathway for the synthesis of 2-alkoxyimidazopyridine.

Scheme 103: Iron-catalyzed synthetic approach.

Scheme 104: Iron-catalyzed aminooxygenation reaction.

Scheme 105: Mechanistic pathway.

Scheme 106: Rh(III)-catalyzed double C–H activation of 2-substituted imidazoles and alkynes.

Scheme 107: Plausible reaction mechanism.

Scheme 108: Rh(III)-catalyzed non-aromatic C(sp2)–H bond activation–functionalization for the synthesis of imid...

Scheme 109: Reactivity and selectivity of different substrates.

Scheme 110: Rh-catalyzed direct C–H alkynylation by Li et al.

Scheme 111: Suggested radical mechanism.

Scheme 112: Scandium(III)triflate-catalyzed one-pot reaction and its mechanism for the synthesis of benzimidazo...

Scheme 113: RuCl3-assisted Ugi-type Groebke–Blackburn condensation reaction.

Scheme 114: C-3 aroylation via Ru-catalyzed two-component reaction.

Scheme 115: Regioselective synthetic mechanism.

Scheme 116: La(III)-catalyzed one-pot GBB reaction.

Scheme 117: Mechanistic approach for the synthesis of imidazo[1,2-a]pyridines.

Scheme 118: Synthesis of imidazo[1,2-a]pyridine using LaMnO3 NPs under neat conditions.

Scheme 119: Mechanistic approach.

Scheme 120: One-pot 3-CR for regioselective synthesis of 2-alkoxy-3-arylimidazo[1,2-a]pyridines.

Scheme 121: Formation of two possible products under optimization of the catalysts.

Scheme 122: Mechanistic strategy for NiFe2O4-catalyzed reaction.

Scheme 123: Two-component reaction for synthesizing imidazodipyridiniums.

Scheme 124: Mechanistic scheme for the synthesis of imidazodipyridiniums.

Scheme 125: CuI-catalyzed arylation of imidazo[1,2-a]pyridines.

Scheme 126: Mechanism for arylation reaction.

Scheme 127: Cupric acetate-catalyzed double carbonylation approach.

Scheme 128: Radical mechanism for double carbonylation of IP.

Scheme 129: C–S bond formation reaction catalyzed by cupric acetate.

Scheme 130: Cupric acetate-catalyzed C-3 formylation approach.

Scheme 131: Control experiments for signifying the role of DMSO and oxygen.

Scheme 132: Mechanism pathway.

Scheme 133: Copper bromide-catalyzed CDC reaction.

Scheme 134: Extension of the substrate scope.

Scheme 135: Plausible radical pathway.

Scheme 136: Transannulation reaction for the synthesis of imidazo[1,5-a]pyridines.

Scheme 137: Plausible reaction pathway for denitrogenative transannulation.

Scheme 138: Cupric acetate-catalyzed C-3 carbonylation reaction.

Scheme 139: Plausible mechanism for regioselective C-3 carbonylation.

Scheme 140: Alkynylation reaction at C-2 of 3H-imidazo[4,5-b]pyridines.

Scheme 141: Two-way mechanism for C-2 alkynylation of 3H-imidazo[4,5-b]pyridines.

Scheme 142: Palladium-catalyzed SCCR approach.

Scheme 143: Palladium-catalyzed Suzuki coupling reaction.

Scheme 144: Reaction mechanism.

Scheme 145: A phosphine free palladium-catalyzed synthesis of C-3 arylated imidazopyridines.

Scheme 146: Palladium-mediated Buchwald–Hartwig cross-coupling reaction.

Figure 7: Structure of the ligands optimized.

Scheme 147: Palladium acetate-catalyzed direct arylation of imidazo[1,2-a]pyridines.

Scheme 148: Palladium acetate-catalyzed mechanistic pathway.

Scheme 149: Palladium acetate-catalyzed regioselective arylation reported by Liu and Zhan.

Scheme 150: Mechanism for selective C-3 arylation of IP.

Scheme 151: Pd(II)-catalyzed alkenylation reaction with styrenes.

Scheme 152: Pd(II)-catalyzed alkenylation reaction with acrylates.

Scheme 153: A two way mechanism.

Scheme 154: Double C–H activation reaction catalyzed by Pd(OAc)2.

Scheme 155: Probable mechanism.

Scheme 156: Palladium-catalyzed decarboxylative coupling.

Scheme 157: Mechanistic cycle for decarboxylative arylation reaction.

Scheme 158: Ligand-free approach for arylation of imidazo[1,2-a]pyridine-3-carboxylic acids.

Scheme 159: Mechanism for ligandless arylation reaction.

Scheme 160: NHC-Pd(II) complex assisted arylation reaction.

Scheme 161: C-3 arylation of imidazo[1,2-a]pyridines with aryl bromides catalyzed by Pd(OAc)2.

Scheme 162: Pd(II)-catalyzed C-3 arylations with aryl tosylates and mesylates.

Scheme 163: CDC reaction for the synthesis of imidazo[1,2-a]pyridines.

Scheme 164: Plausible reaction mechanism for Pd(OAc)2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 165: Pd-catalyzed C–H amination reaction.

Scheme 166: Mechanism for C–H amination reaction.

Scheme 167: One-pot synthesis for 3,6-di- or 2,3,6-tri(hetero)arylimidazo[1,2-a]pyridines.

Scheme 168: C–H/C–H cross-coupling reaction of IPs and azoles catalyzed by Pd(II).

Scheme 169: Mechanistic cycle.

Scheme 170: Rh-catalyzed C–H arylation reaction.

Scheme 171: Mechanistic pathway for C–H arylation of imidazo[1,2-a]pyridine.

Scheme 172: Rh(III)-catalyzed double C–H activation of 2-phenylimidazo[1,2-a]pyridines and alkynes.

Scheme 173: Rh(III)-catalyzed mechanistic pathway.

Scheme 174: Rh(III)-mediated oxidative coupling reaction.

Scheme 175: Reactions showing functionalization of the product obtained by the group of Kotla.

Scheme 176: Mechanism for Rh(III)-catalyzed oxidative coupling reaction.

Scheme 177: Rh(III)-catalyzed C–H activation reaction.

Scheme 178: Mechanistic cycle.

Scheme 179: Annulation reactions of 2-arylimidazo[1,2-a]pyridines and alkynes.

Scheme 180: Two-way reaction mechanism for annulations reaction.

Scheme 181: [RuCl2(p-cymene)]2-catalyzed C–C bond formation reaction.

Scheme 182: Reported reaction mechanism.

Scheme 183: Fe(III) catalyzed C-3 formylation approach.

Scheme 184: SET mechanism-catalyzed by Fe(III).

Scheme 185: Ni(dpp)Cl2-catalyzed KTC coupling.

Scheme 186: Pd-catalyzed SM coupling.

Scheme 187: Vanadium-catalyzed coupling of IP and NMO.

Scheme 188: Mechanistic cycle.

Scheme 189: Selective C3/C5–H bond functionalizations by mono and bimetallic systems.

Scheme 190: rGO-Ni@Pd-catalyzed C–H bond arylation of imidazo[1,2-a]pyridine.

Scheme 191: Mechanistic pathway for heterogeneously catalyzed arylation reaction.

Scheme 192: Zinc triflate-catalyzed coupling reaction of substituted propargyl alcohols.
Mechanochemistry of supramolecules
- Anima Bose and
- Prasenjit Mal
Beilstein J. Org. Chem. 2019, 15, 881–900, doi:10.3762/bjoc.15.86
- , Figure 4b). Contrastingly, in solution the same synthesis took 4 h at 70 °C to isolate complex 9 in 56% yield. Ćurić and co-workers prepared cyclopalladated complexes such as 10 by a grinding method and were the first to confirm a mechanochemical C–H bond activation of an unsymmetrically substituted
- salen-type ligand followed by metal-complex formation in the same pot [55]. Otera’s solvent-free approach by which the formation of self-assembled supramolecules could be accelerated [56]. Synthesis of a Pd-based metalla-supramolecular assembly through mechanochemical activation for C–H-bond activation

Graphical Abstract

Figure 1: A generalized overview of coordination-driven self-assembly.

Figure 2: Examples of self-assembly or self-sorting and subsequent substitution.

Figure 3: Synthesis of salen-type ligand followed by metal-complex formation in the same pot [55].

Figure 4: Otera’s solvent-free approach by which the formation of self-assembled supramolecules could be acce...

Figure 5: Synthesis of a Pd-based metalla-supramolecular assembly through mechanochemical activation for C–H-...

Figure 6: a) Schematic representation for the construction of a [2]rotaxane. b) Chiu’s ball-milling approach ...

Figure 7: Mechanochemical synthesis of the smallest [2]rotaxane.

Figure 8: Solvent-free mechanochemical synthesis of pillar[5]arene-containing [2]rotaxanes [61].

Figure 9: Mechanochemical liquid-assisted one-pot two-step synthesis of [2]rotaxanes under high-speed vibrati...

Figure 10: Mechanochemical (ball-milling) synthesis of molecular sphere-like nanostructures [63].

Figure 11: High-speed vibration milling (HSVM) synthesis of boronic ester cages of type 22 [64].

Figure 12: Mechanochemical synthesis of borasiloxane-based macrocycles.

Figure 13: Mechanochemical synthesis of 2-dimensional aromatic polyamides.

Figure 14: Nitschke’s tetrahedral Fe(II) cage 25.

Figure 15: Mechanochemical one-pot synthesis of the 22-component [Fe4(AD2)6]4− 26, 11-component [Fe2(BD2)3]2− ...

Figure 16: a) Subcomponent synthesis of catalyst and reagent and b) followed by multicomponent reaction for sy...

Figure 17: A dynamic combinatorial library (DCL) could be self-sorted to two distinct products.

Figure 18: Mechanochemical synthesis of dynamic covalent systems via thermodynamic control.

Figure 19: Preferential formation of hexamer 33 under mechanochemical shaking via non-covalent interactions of...

Figure 20: Anion templated mechanochemical synthesis of macrocycles cycHC[n] by validating the concept of dyna...

Figure 21: Hydrogen-bond-assisted [2 + 2]-cycloaddition reaction through solid-state grinding. Hydrogen-bond d...

Figure 22: Formation of the cage and encapsulation of [2.2]paracyclophane guest molecule in the cage was done ...

Figure 23: Formation of the 1:1 complex C60–tert-butylcalix[4]azulene through mortar and pestle grinding of th...

Figure 24: Formation of a 2:2 complex between the supramolecular catalyst and the reagent in the transition st...

Figure 25: Halogen-bonded co-crystals via a) I···P, b) I···As, and c) I···Sb bonds [112].

Figure 26: Transformation of contact-explosive primary amines and iodine(III) into a successful chemical react...

Figure 27: Undirected C–H functionalization by using the acidic hydrogen to control basicity of the amines [114]. a...






